Written by: Mike Tandlich, MD (NUEM ‘24) Edited by: Peter Serina, MD, MPH (NUEM ‘22)
Expert Commentary by: Mike Macias, MD (NUEM ‘17)
Expert Commentary
Thank you Drs. Tandlich and Serina for this excellent infographic summarizing stingray envenomation! The good news is that the majority of stingray injuries are nonfatal and will heal without any complications! You hit all of the key points however I just wanted to highlight a few management tips below:
Treat as a Trauma!
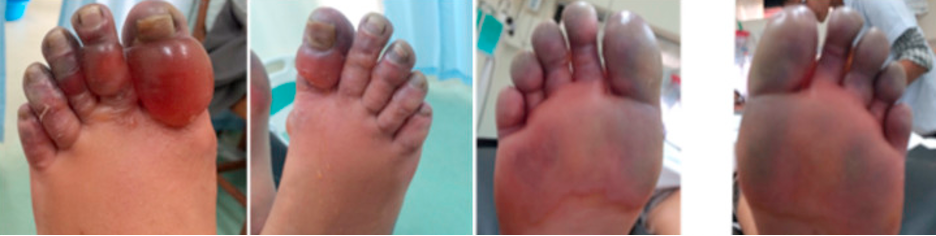
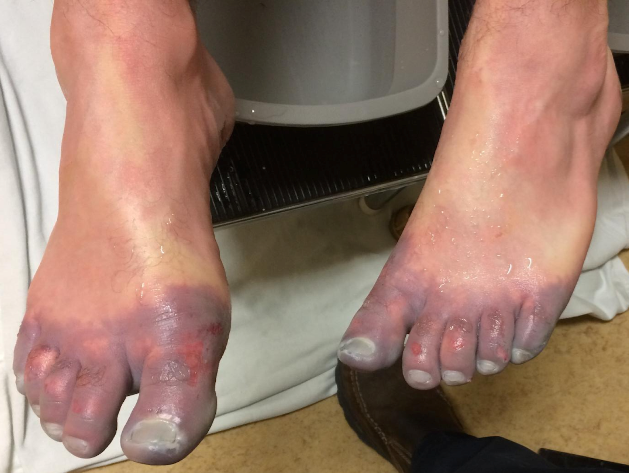
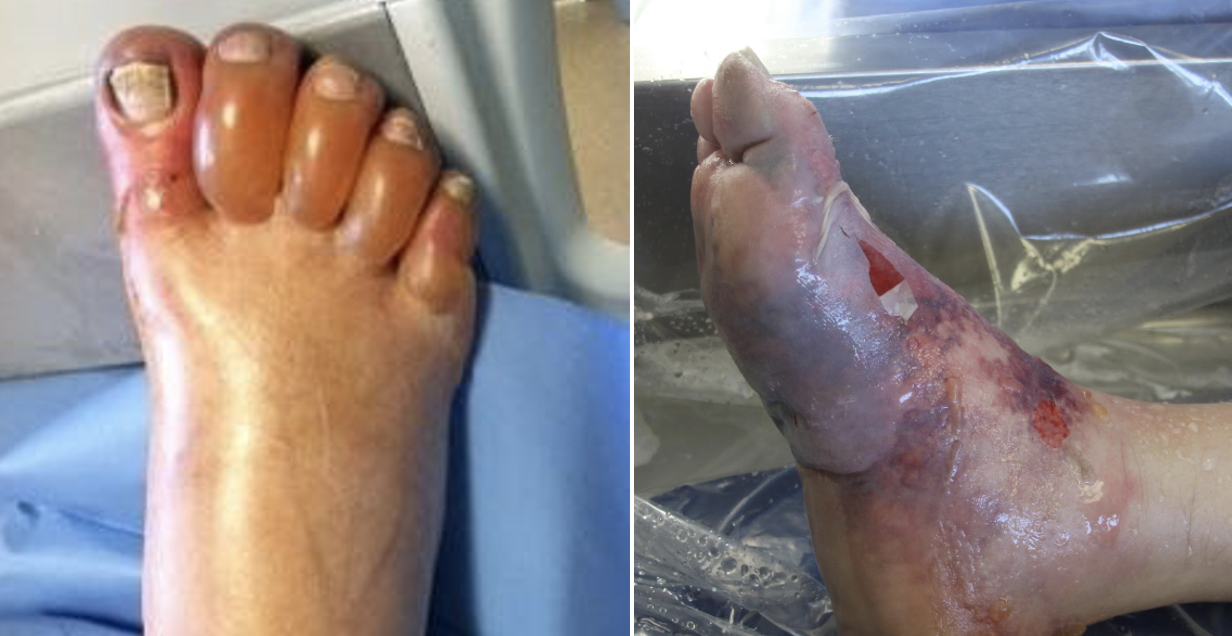
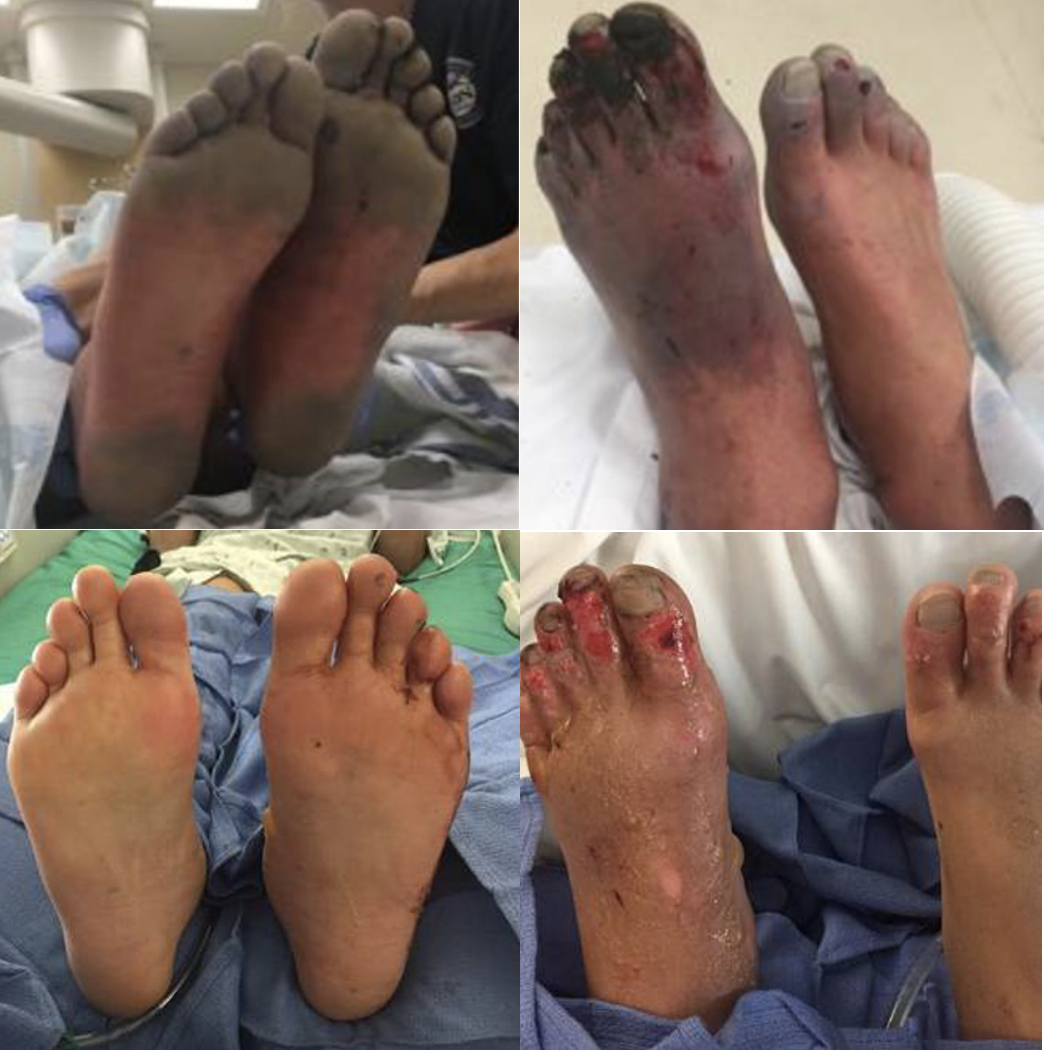
While majority of the pain from stingray envenomation occurs as a result of its venom, it is important to remember that this is also a traumatic injury. Treat the injury just like you would any other penetrating trauma. Consider the location as well as surrounding structures and make sure to properly examine for tendon, nerve, and vascular injury. Injuries to the chest or abdominal regions should prompt advanced imaging and trauma consultation.
Hot Water is Key!
Stingray envenomation is noted to cause severe pain that is often out of proportion to your examination findings. While the exact mechanism is not clear, the venom can lead to not only pain but also local tissue necrosis. The good news is the venom is heat labile! The faster you can get the injured area into hot water the better. You want the water to be as hot as tolerable without causing a thermal burn. A good rule of thumb is to have the patient place their unaffected limb in the water first to see if it is tolerable. As this often occurs at a beach, lifeguards are often your best resource to get hot water fast! Oral analgesics can be administered if needed however often they are unnecessary as soon as the injured area is submerged in hot water.
Retained Barb?
While uncommon, a retained barb from the envenomation can occur so be sure to consider this and evaluate appropriately. Traditionally, x-ray imaging of the affected area is performed to evaluate for a radio-opaque barb however some evidence suggests this to be a relatively low yield practice [1]. Ultrasound can also be considered if there is suspicion for retained barb or other material. In general ultrasound has been shown to be highly sensitive for identification of foreign body [2]. Not only can it be used to identify the barb but it can be used to facilitate removal [3].
Give Prophylactic Antibiotics
Prophylactic antibiotics are recommended for stingray envenomation given that the limited data suggest a higher rate of wound infection in patients who were not initially treated with antibiotics [1]. Given these injuries often occur in the ocean make sure to cover for salt water species such as Vibro. Levofloxacin is my go to option.
Teach The Stingray Shuffle!
Keeping these key management points in mind, the good news is that the majority of stingray injuries are nonfatal and will heal without any complications! Before your patient is discharged don’t forget to remind them that the next time they are going out for a surf to do the stingray shuffle!
References
Clark RF, Girard RH, Rao D, Ly BT, Davis DP. Stingray envenomation: a retrospective review of clinical presentation and treatment in 119 cases. J Emerg Med. 2007 Jul;33(1):33-7
Aras MH, Miloglu O, Barutcugil C, Kantarci M, Ozcan E, Harorli A. Comparison of the sensitivity for detecting foreign bodies among conventional plain radiography, computed tomography and ultrasonography. Dentomaxillofac Radiol. 2010;39(2):72-78. doi:10.1259/dmfr/68589458
Nwawka OK, Kabutey NK, Locke CM, Castro-Aragon I, Kim D. Ultrasound-guided needle localization to aid foreign body removal in pediatric patients. J Foot Ankle Surg. 2014;53(1):67-70. doi:10.1053/j.jfas.2013.09.006
Michael Macias, MD
Systems Clinical Ultrasound Director,
Emergent Medical Associates
Ultrasound Director,
UHS SoCal MEC Residency Programs
How To Cite This Post:
[Peer-Reviewed, Web Publication] Tandlich, M. Serina, P. (2021, Nov 15). Stingray Stings. [NUEM Blog. Expert Commentary by Macias, M]. Retrieved from http://www.nuemblog.com/blog/stingray-stings


















































































![Figure 1: Composition of Various Brands of Lipid Emulsions[1]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1594923133383-9YSM79RGG2VRCXN045S4/Intralipid+image+1.png)
![Figure 2: Maximum Doses and Durations of Various Local Anesthetics[9]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1595368949352-3SYZ5X29MWCMBS3CV8BT/Screen+Shot+2020-07-21+at+5.02.19+PM.png)