A case discussion and exploration of the mechanisms and complications of Kratom use
Neuroleptic Malignant Syndrome
Written by: Maren Leibowitz, MD (NUEM ‘23) Edited by: Nick Wleklinski, MD (NUEM ‘22)
Expert Commentary by: Zachary Schmitz, MD (NUEM '21)
Expert Commentary
This is an awesome, focused review of neuroleptic malignant syndrome (NMS). NMS is hard to diagnose because it's rare. There is no gold standard with respect to its definition, and it requires a medication history (which we typically don't do very well in the emergency department). A tricky cause of NMS is the removal of a dopamine agonist. For this reason, carbidopa/levodopa should never be discontinued during hospital admission - or ED boarding. [1]
Supportive care is more important than antidotal therapy during NMS management. The most acute cause of death from NMS is hyperthermia, which is induced both by D2 receptor antagonism leading to rigidity and impaired thermoregulation from the striatum and hypothalamus. Any life-threatening hyperthermia should be treated immediately with an ice bath.[2] Rigidity will lead to rhabdomyolysis with subsequent hyperkalemia and myoglobin-induced renal failure. Therefore, fluid resuscitation and maintenance are important. Profound immobility can precipitate DVT, so anticoagulation may be necessary.
In terms of pharmacotherapy, benzodiazepines are universally used. Dantrolene inhibits calcium-mediated muscle contraction to reduce muscle rigidity. However, it doesn't address the underlying central D2 antagonism, and its efficacy has only been shown in case reports. Bromocriptine acts more centrally as a dopamine agonist but should be used cautiously in patients with psychiatric diseases as it may exacerbate psychosis. Overall, benzodiazepine use and supportive care should get you through most cases of NMS, though additional therapies may be necessary in severe cases.
References
1. Institute for Safe Medication Practices. Delayed Administration and Contraindicated Drugs Place Hospitalized Parkinson’s Disease Patients at Risk. 12 March 2015. Accessed February 11, 2022.
2. Juurlink JN. Antipsychotics. In: Nelson LS, Howland M, Lewin NA, Smith SW, Goldfrank LR, Hoffman RS. eds. Goldfrank's Toxicologic Emergencies, 11e. Page 1037-1039. McGraw Hill; 2019. Accessed February 11, 2022.
Zachary Schmitz, MD
Toxicology Fellow
Ronald O. Perelman Department of Emergency Medicine
NYU Langone Health
How To Cite This Post:
[Peer-Reviewed, Web Publication] Leibowitz, M. Wleklinski, N. (2022, May 9). Neuroleptic Malignant Syndrome. [NUEM Blog. Expert Commentary by Schmitz, Z]. Retrieved from http://www.nuemblog.com/blog/neuroleptic-malignant-syndrome.
Other Posts You May Enjoy
Beyond the Burns: Toxic House Fire Gases
Written by: Adam Payne, MD (NUEM ‘24) Edited by: Emily Wessling, MD (NUEM ‘22)
Expert Commentary by: Justin Seltzer, MD (NUEM ‘21)
Expert Commentary
Congratulations to Drs. Payne and Wessling on an excellent post.
Management of toxic gas exposure from a house fire is essential knowledge for all emergency physicians. The two major toxic gases of interest are carbon monoxide and hydrogen cyanide; it is important to note that fires in other environments, such as factories or industrial sites, may result in alternative exposures based on the nature of the fire and materials present. The post goes into detail on the pathophysiology, signs, symptoms, and diagnosis of both carbon monoxide and cyanide poisonings, so this commentary will focus on the clinical approach.
I recommend the following simplified process to streamline diagnosis and treatment decision making:
1. Start high flow oxygen (15L NRB) immediately (or, if intubated, give 100% FiO2)
High flow oxygen reduces the half life of carbon monoxide from ~5 hours to ~90 minutes. Oxygen can be discontinued once carboxyhemoglobin normalizes (<2%)
2. Obtain a carboxyhemoglobin level, lactic acid level, and a venous blood gas (arterial is unnecessary unless oxygenation is also a concern) as soon as possible
Unfortunately, there is some disagreement as to what constitutes a “toxic” carboxyhemoglobin level. Weaver, et al. established 25% as an inflection point for the development of severe sequelae, which is now a commonly used (though not universally agreed upon) threshold value. Also, when evaluating a carboxyhemoglobin level, it is essential to consider the time period prior to the level being drawn to avoid false reassurance. For example, a level drawn after 90 minutes of high flow oxygen will be reduced by roughly 50% and interpreted accordingly. Further, do not rely on external co-oximeters alone to rule out carbon monoxide poisoning given limited sensitivity at the moment (though the technology will likely improve over time).
3. Treat with hydroxocobalamin empirically if symptomatic and/or if lactic acid elevated
Hydroxocobalamin is a low risk intervention with significant potential therapeutic benefit, so it should be given early if there is any clinical concern. The other major cyanide antidote, sodium thiosulfate, should only be used if hydroxocobalamin is not available as it has no efficacy advantage and an undesirable side effect profile. It is essential to avoid using nitrites, as inducing methemoglobinemia in the setting of coincidental carbon monoxide poisoning can dramatically worsen tissue hypoxia.
An elevated lactic acid is a surrogate for cyanide poisoning, specifically a level of 8-10 mmol/L or greater is sensitive and should prompt intervention. More modest lactic acid elevations are less likely to be related to cyanide poisoning and should not prompt intervention, especially in an asymptomatic patient, unless the level is persistently elevated despite adequate resuscitation. Cyanide levels, while diagnostic, are of no acute clinical utility since they are rarely available in a timely manner.
4. Consider hyperbaric oxygen therapy (HBOT) if readily available
HBOT for the treatment of carbon monoxide poisoning is controversial. While HBOT reduces the half life of carbon monoxide to roughly 20-30 minutes, HBOT is not used for this purpose alone. In fact, HBOT is primarily used to reduce associated cognitive, behavioral, and neurologic changes (collectively known as delayed neuropsychiatric sequelae). There is lower quality evidence for reduced myocardial infarction and mortality risk as well.
However, several factors limit HBOT use. Primarily, it is not universally available at most institutions, incurring the risk and cost of transport to a distant site. The benefit is thought to be highest when the treatment is performed early (ideally within six hours of exposure), which adds to the logistical burden. Additionally, many chambers are not operated on nights and weekends, and of those available at off hours, many are unable to accommodate intubated patients. Recognizing the controversial nature of HBOT, the 2016 ACEP position statement noted that HBOT or high flow, normobaric oxygen can be used to treat carbon monoxide poisoning; though it likely carries clinical benefit in certain situations, at this time HBOT is not the standard of care for severe carbon monoxide poisoning. Rather, it should be offered if it can be readily and reasonably arranged.
Keeping this in mind, the following are generally accepted indications for HBOT in the setting of carbon monoxide poisoning
Loss of consciousness associated with exposure
Altered mental status
Focal neurologic changes
Evidence of end organ ischemia (pH ≤ 7.1, EKG changes, elevated troponin, angina)
Pregnancy (with some resources citing a level ≥20%)
Carboxyhemoglobin level ≥25%
Importantly, these are not hard and fast rules and there is no firm guideline mandating when HBOT should or should not be used. As a result, it is prudent to involve a medical toxicologist early in the process.
In summary, a few key take home points:
Carbon monoxide and cyanide are strongly associated with house fires – assume exposure to both until proven otherwise
It is reasonable to treat any undifferentiated, symptomatic patient with high flow oxygen and hydroxocobalamin empirically; asymptomatic patients can wait safely for blood work on high flow oxygen alone
The decision making regarding use of HBOT, in particular, is complex – early consultation with a medical toxicologist is strongly encouraged
Justin Seltzer, MD
Toxicology Fellow
Department of Emergency Medicine
University of California, San Diego
How To Cite This Post:
[Peer-Reviewed, Web Publication] Payne, A. Wessling, E. (2022, May 2). Toxic House Fire Gases. [NUEM Blog. Expert Commentary by Seltzer, J]. Retrieved from http://www.nuemblog.com/blog/toxic-house-fire-gases
Other Posts You May Enjoy
Toxic Alcohols
Written by: Rafael Lima, MD (NUEM ‘23) Edited by: Laurie Aluce, MD (NUEM ‘21)
Expert Commentary by: Zachary Schmitz (NUEM ‘21)
Methanol Toxicity
Methanol itself is not toxic to the body. Methanol’s metabolite, formic acid, causes toxicity at serum levels greater than 20mg/dl [1].
Clinical Findings of Methanol Poisoning
CNS sedation
Seizures
Rapid, Deep Breathing
Hypotension
Ocular findings:
Blindness
Afferent pupillary defect
Optic disk hyperemia
Mydriasis
Ethylene Glycol Toxicity
Similarly, the toxic metabolites of ethylene glycol cause end-organ damage at levels greater than 20mg/dl. The most notable toxic metabolites are glycolic acid and oxalic acid.” [1] .
Clinical Findings of Ethylene Glycol Poisoning
CNS sedation
Seizures
Cranial nerve palsies
Rapid, deep breathing
Hypotension
Hypocalcemia (can result in tetany)
Renal findings:
Oliguria
Acute renal failure
Flank pain
Hematuria
Oxalate crystals in the urine under fluorescence
Isopropyl Alcohol Toxicity
Found in hand sanitizers and disinfectants, isopropyl alcohol is a less common source of alcohol poisoning. The parent molecule does exhibit toxic effects here, unlike methanol and ethylene glycol. If untreated, the lethal dose is between 4-8 g/kg [2].
Alcohol dehydrogenase metabolizes isopropyl alcohol into acetone. Because acetone is a ketone, and ketones are not oxidized into carboxylic acids, isopropyl alcohol poisoning does not result in anion gap metabolic acidosis.
Clinical Findings of Isopropyl Alcohol Poisoning
CNS sedation
Disconjugate gaze
Fruity breath odor
Hypotension
Hematemesis
Pulmonary edema
Plasma Osmolal Gap
One of the most reliable laboratory markers of toxic alcohol poisoning is a large osmolal gap. The osmolal gap is defined as the difference between the measured serum osmolality and the calculated, or expected, plasma osmalality:
OSMOLAR GAP = Measured plasma osmolality – calculated/expected plasma osmolality
The common equation for calculating the expected plasma osmolality is listed below [3]. Of note, there are other formulas with slight variations. Using an online calculator can be helpful.
Expected Serum Osmolality=2[Na]+BUN/2.8+Glucose/18
A gap < 10 is considered normal. Any elevation above 10 should raise the clinician’s suspicion of toxic alcohol ingestion.
Note: this tool is not helpful in late presentations as the metabolized forms of the different alcohols do not contribute to the osmolal gap. The calculated gap will be falsely low in late-stage poisoning.
Treatment of Toxic Alcohol Ingestions
Consult your medical toxicologist or poison control center if toxic alcohol ingestion is suspected.
The national poison control center hotline telephone number is 1(800)-222-1222.
Fomepizole
Fomepizole should be used only for methanol and ethylene glycol ingestions. It is not indicated for isopropyl alcohol intoxications [4]. It is an inhibitor of alcohol dehydrogenase (ADH). Evidence shows that it is a superior antidote to ethanol [5].
Loading dose 15 mg/kg IV
Then 10 mg/kg every 12 hours
Continue until blood pH is normal and serum alcohol concentration is less than 20 mg/dL in the presence of retinal or renal injury.
Ethanol
Ethanol works as a competitive inhibitor of ADH, having a higher affinity for the enzyme compared to the other alcohols. Ethanol was used historically before the effects of fomepizole were studied. Fomepizole is now the preferred treatment because the administration of ethanol is more difficult, ethanol causes sedation, and titration of the therapy is challenging in co-ingestions [6]. If ethanol must be used, the preferred route is IV and the studied therapeutic target level is 100 mg/dL [7].
Supplemental Therapy
Methanol poisoning patients should also receive folic acid (50mg IV every 6 hours) [7].
Ethylene glycol poisoning patients should also receive thiamine (100mg IV) and pyridoxine (50mg IV) [8].
Hemodialysis
Consult your nephrologist early if you are considering hemodialysis. Renal replacement therapy should be considered in the following situations [9]:
Anion gap metabolic acidosis with known toxic alcohol ingestion
End-organ damage
Renal failure
Vision changes
Unexplained anion gap metabolic acidosis with elevated osmolal gap in suspected toxic alcohol ingestion
References
1. Liesivuori, J. and H. Savolainen, Methanol and formic acid toxicity: biochemical mechanisms. Pharmacol Toxicol, 1991. 69(3): p. 157-63.
2. Slaughter, R.J., et al., Isopropanol poisoning. Clin Toxicol (Phila), 2014. 52(5): p. 470-8.
3. Bhagat, C.I., et al., Calculated vs measured plasma osmolalities revisited. Clin Chem, 1984. 30(10): p. 1703-5.
4. Su, M., R.S. Hoffman, and L.S. Nelson, Error in an emergency medicine textbook: isopropyl alcohol toxicity. Acad Emerg Med, 2002. 9(2): p. 175.
5. McMartin, K., D. Jacobsen, and K.E. Hovda, Antidotes for poisoning by alcohols that form toxic metabolites. Br J Clin Pharmacol, 2016. 81(3): p. 505-15.
6. Zakharov, S., et al., Fomepizole versus ethanol in the treatment of acute methanol poisoning: Comparison of clinical effectiveness in a mass poisoning outbreak. Clin Toxicol (Phila), 2015. 53(8): p. 797-806.
7. Barceloux, D.G., et al., American Academy of Clinical Toxicology practice guidelines on the treatment of methanol poisoning. J Toxicol Clin Toxicol, 2002. 40(4): p. 415-46.
8. Ghosh, A. and R. Boyd, Leucovorin (calcium folinate) in "antifreeze" poisoning. Emerg Med J, 2003. 20(5): p. 466.
9. Moreau, C.L., et al., Glycolate kinetics and hemodialysis clearance in ethylene glycol poisoning. META Study Group. J Toxicol Clin Toxicol, 1998. 36(7): p. 659-66.
Expert Commentary
Thank you for this great review of a difficult subject! The combination of a lack of quick, confirmatory testing with delayed onset of symptoms makes toxic alcohol poisoning an incredibly difficult diagnosis to make. Additionally, even small ingestion can lead to major complications. For example, if a typical four-year-old (19kg) child drank windshield washer fluid that contained 50% methanol (a fairly standard formulation), it would take only 5.7 mL to potentially produce a methanol serum concentration of 25 mg/dL. Given the average 4-year-old’s mouthful is 8.9 mL, you can run into trouble quickly.[1]
We frequently see misuse or misunderstanding of osmol and anion gaps in diagnosing toxic alcohol ingestion when history is unclear. First, although a normal osmol gap is generally less than 10, baseline osmol gaps range from -10 to +14.[2] Therefore, a gap of 16 may represent a true gap of +2 in one person and +26 in another. Second, ethanol must be included in the osmol gap equation. An ethanol concentration of 200 mg/dL would increase your osmol gap by 43.5. Third, given metabolism over time, all values included in an anion gap calculation need to be drawn off of the same blood sample.
These considerations make finding the diagnosis even more complicated, but there are a few things that can help you out. First, an osmol gap > 50 is highly concerning for toxic alcohol. Second, an ethanol concentration > 100 mg/dL is sufficient to block ADH, meaning that few toxic metabolites from methanol or ethylene glycol could be made.[3] This means that an anion gap present with an ethanol > 100 mg/dL is not from toxic alcohol (unless the patient drank the ethanol after the toxic alcohol, which is very rare). Third, sequential values over time can be helpful. Metabolism of toxic alcohols should lead to a decreased osmol gap and increased anion gap over time. Proper use of the osmol and anion gap can help identify patients at high risk for morbidity and mortality while decreasing unnecessary administration of fomepizole, which typically costs thousands of dollars.
References
Ratnapalan S, Potylitsina Y, Tan LH, Roifman M, Koren G. Measuring a toddler's mouthful: toxicologic considerations. Journal of Pediatrics. 2003 Jun;142(6):729-30. doi: 10.1067/mpd.2003.216
Hoffman RS, Smilkstein MJ, Howland MA, Goldfrank LR. Osmol gaps revisited: normal values and limitations. J Toxicol Clin Toxicol. 1993;31(1):81-93. doi: 10.3109/15563659309000375.
Jacobsen D, McMartin KE. Methanol and ethylene glycol poisonings: mechanism of toxicity, clinical course, diagnosis and treatment. Med Toxicol. 1986;1:309-334.
Zachary Schmitz, MD
Toxicology Fellow
Ronald O. Perelman Department of Emergency Medicine
NYU Langone Health
How To Cite This Post:
[Peer-Reviewed, Web Publication] Lima, R. Aluce, L. (2022, Jan 24). Toxic Alcohols. [NUEM Blog. Expert Commentary by Schmitz, Z]. Retrieved from http://www.nuemblog.com/blog/toxic-alcohols
Other Posts You May Enjoy
Marine Envenomations
Written by: Michael Tandlich, MD (NUEM ‘24) Edited by: Chloe Renshaw, MD (NUEM ‘22)
Expert Commentary by: Justin Seltzer, MD (NUEM ‘21)
Expert Commentary
An excellent post by Drs. Tandlich and Renshaw. Marine envenomations are common problems around the world. Like with land-based envenomations, the venomous organisms of note vary with geography; jellyfish encountered in Australia are different from those encountered in Florida, for example. As a result, we will focus on major envenomations in the United States.
The invertebrates account for a large but ultimately unknown number of envenomations. Cnidaria includes jellyfish, hydrozoa, anemones, and fire coral. A majority of stings from this group result in painful dermatitis; tentacles create a “whip-like” pattern on the skin, whereas fire coral creates localized skin wheals. The sea nettle and Portuguese man-of-war are of greatest interest, given their potential to cause severe systemic symptoms. Box jellyfish are rare in US coastal waters but produce a life-threatening toxicity.
Initial treatment is somewhat controversial. Many resources advocate for the use of seawater for the initial decontamination, given concern for vinegar triggering nematocyst release in some species common to US waters. However, further research is needed to determine which is best. At this time, seawater is recommended for empiric decontamination in the US unless a box jellyfish is strongly suspected, in which case vinegar is appropriate (a very rare circumstance). Systemically ill box jellyfish envenomations should be treated with pain and blood pressure control. The antivenom is not readily available in the US and is unlikely to be beneficial in the time course it would take to obtain it.
Echinodermata, which includes sea urchins, have mild venom on their spines that can cause local tissue irritation and pain. There are reports of severe envenomations with systemic symptoms, but this is ultimately quite rare. These injuries respond well to hot water immersion. Imaging and local wound exploration for retained spines are recommended. Soaking the wound in vinegar may help dissolve superficial spines.
Of the vertebrates, stingrays and spiny fish are of primary concern.
Stingrays stings are common and can cause serious penetrating trauma but envenomation mainly produces localized pain and swelling. The venom is heat-labile, so significant pain relief can be achieved with hot water immersion. Stingrays stings have the potential for both retained stinger and wound infections; evaluation for retained stinger with radiographs and local wound exploration is recommended along with prophylactic antibiotics.
Spinyfish, in particular stonefish, lionfish, and scorpionfish, have venom located in their spines. Stonefish have the most potent venom of any known fish. Lionfish are not native to the US but have become an invasive species. Human contact with these fish occurs both in the wild and in aquariums. These fish also have heat-labile venom susceptible to hot water immersion. However, systemically ill stonefish envenomations should receive the antivenom as this envenomation can be life-threatening; the antivenom will likely work against other spiny fish too, however, these other envenomations are usually much less severe and rarely require more than hot water immersion and supportive care.
So key learning points:
Most marine envenomations involve heat-labile venom. Hot water immersion is likely to help reduce local symptoms.
Systemic illness is rare but some marine envenomations can produce life-threatening toxicity. Be very wary of a systemically ill envenomation and try to figure out the source due to the limited availability of antivenoms.
Prophylactic antibiotics are recommended for stingray stings as they tend to get infected but otherwise are generally not necessary in most populations. Good wound care, evaluation for retained foreign bodies, and tetanus prophylaxis are the mainstays.
For further information, see this review article
Justin Seltzer, MD
UCSD Health Toxicology Fellow
Emergency Physician, UCSD Health
How To Cite This Post:
[Peer-Reviewed, Web Publication] Tandlich, M. Renshaw, C. (2022, Mar 7). Marine Envenomations. [NUEM Blog. Expert Commentary by Seltzer, J]. Retrieved from http://www.nuemblog.com/blog/marine-envenomations
Other Posts You May Enjoy
Management of Snake Bite Injuries
Written by: Rafael Lima, MD (NUEM ‘23) Edited by: Mike Conrardy, MD (NUEM ‘21) Expert Commentary by: Sean Bryant, MD
An estimated 10,000 patients visit emergency departments for snake bite injuries each year in the United States [1]. The number of snake bite occurrences an emergency department sees depends largely on the geographic area of practice. While there are known remedies for these incidents, snake bites can be devastating if not promptly managed, meaning emergency physicians should be knowledgeable in the subject. In this article, we review the common management of snake bite injuries and envenomations for the two major snake groups in the United States.
Overview
There are about 20 known venomous species of snakes in the United States. While most envenomations occur in the Southwestern United States, every region is home to at least one species of venomous snake [2]. Not all snake bites result in envenomation. At least 25% of venomous snake bites are dry. You should still suspect envenomation upon the patient’s initial presentation and rule it out by monitoring their clinical symptoms and progression.
Identification of the snake is useful in guiding management of care, but it should not be attempted if doing so poses any additional risk to the patient or provider. In the United States, venomous snakes generally fall under two categories: Crotaline/pit vipers in the Viperidae family, and coral snakes in the Elapidae family.
Crotaline (Pit Vipers)
This group of snakes has historically been responsible for the more severe envenomations between the two groups [3]. The WHO classifies pit vipers in CAT 1 of their venom database, describing them as highly venomous with high rates of morbidity and mortality [4].
Pit vipers generally have a triangular shaped head with heat-sensing “pits” located on the face. They frequently have a rattle on their tail, but not all pit vipers are rattlesnakes. Copperheads and cottonmouth snakes are also included in this group.
Crotaline venom causes localized tissue necrosis and congestive coagulopathy. This can be identified by a prolonged INR, PT, PTT, and thrombocytopenia. Additionally, the viper venom can cause capillary and cellular membranes to increase in permeability. Large amounts of venom can cause diffuse vaso-extravasation and hemolysis that can lead to hypovolemic shock and DIC if untreated.
CroFab is the antivenom of choice for cotaline envenomation. It is a polyvalent antivenom, meaning it contains antibodies derived from the venom of multiple different species of snakes. Administration is titrated based on clinical and symptom response.
Elapidae
The venomous Elapidae snake in the United States is the coral snake. There are less severe envenomations from coral snakes compared to pit vipers. This is a result of how venom is administered between the two groups: pit vipers have venom glands that inject venom directly through the fangs, while coral snakes rely on passive seeping of venom through their glands while they chew.
Source: Tad Arensmeier from St. Louis, MO, USA
Coral snakes can be identified by their brightly colored rings extending along the length of the whole body. Usually, every other ring is yellow, separating the wider red or black rings in between. The common saying “red on yellow, kill a fellow; red on black, venom lack” has been been used to differentiate between venomous coral snakes and their harmless look-alikes in North America. A further level of differentiation is how far the rings extend circumferentially around the snake. Rings encircle the entire body in venomous coral snakes, while harmless look-alikes do not have the red coloration on the ventral side [5].
Source: Dawson at English Wikipedia
Venomous bites by coral snakes usually elicit little to no pain. This is because the Elapidae venom acts upon the neuromuscular junction and inhibits acetylcholine receptors. Clinical manifestations are predominantly neurological. Envenomation can cause lethargy, confusion, salivation, cranial nerve palsies, and respiratory paralysis. Symptoms are usually delayed, up to 12 hours from the initial bite. Coagulopathy and tissue necrosis does not happen with coral snake venom [2]. Unfortunately, the Elapidae antivenom is no longer manufactured in the United States and there is a limited supply available.
ED Work Up
As in all patients who present to the emergency department, first ensure that airway, breathing, and circulation are intact. All suspected snake bite injuries warrant a prompt toxicology or poison center consult.
Sometimes, patients will bring in a dead or decapitated snake for identification in the emergency department. DO NOT attempt to handle a snake the patient brought in for identification, even if it is dead. Many snakes have intact reflexes that are preserved even after death or decapitation and you can still be bitten and envenomated by a dead snake!
Examine the injury and look for clear fang marks or puncture wounds. Get a history focused on the timing of the injury, medication allergies, and description of the snake, if known. The borders of erythema should be measured and marked serially.
Laboratory work-up is focused on assessing coagulopathy and hemolysis, especially if the snake is a confirmed pit viper or is unknown. Obtain CBC with platelet count, PT, PTT, INR, fibrinogen, and D-dimer. It is also important to check a baseline set of electrolytes with a basic chem panel, assess the extent of myonecrosis with a CK, and assess for renal damage with a UA.
Manage the wound with copious irrigation and exploration for retained foreign bodies (ie. fangs or teeth). Inquire about the patient’s tetanus status and administer if they are not up to date. Do not attempt to tourniquet or suction venom out of the wound. There is no evidence for routine antibiotic use in snake injuries [6].
Crotaline Bite Management
Consider using CroFab antivenom if the local area of injury and erythema is expanding. If coagulopathy is detected, do not treat with heparin or FFP. Give antivenom first, as unneutralized venom will react with clotting factor replacements [2]. Patients with abnormal coagulation studies within 12 hours after CroFab administration are more likely to develop recurrent coagulopathy. In these patients, repeat coagulation studies should be obtained every 48 hours until resolved. If lab values are worsening, then antivenom retreatment should be reconsidered [7].
Observe the affected limb for compartment syndrome. If clinical suspicion is high for compartment syndrome, consider formally measuring compartment pressures. Elevate the affected limb, and administer extra vials of antivenom. Antivenom administration is preferred over fasciotomy in the treatment of compartment syndrome caused by Crotaline venom [8].
Crofab, the Crotaline antivenom, is typically administered in stepwise fashion and is titrated to clinical resolution of symptoms. Administer 4-6 vials of CroFab antivenom and watch for clinical improvement at the local site of injury. If no improvement seen, administer 4-6 more vials. Repeat until control is achieved, meaning a reversal of symptoms, such as erythema, swelling, pain. Then administer 2 vial doses 6 hours later, then 12 hours, then 18 hours. Envenomation patients should be monitored for at least 8 hours. Keep epinephrine and antihistamines nearby in case of anaphylaxis or allergy to antivenom [2].
Elapidae Bite Management
Because of their potential devastating neurologic effects, coral snake bites should be empirically treated with antivenom and monitored for respiratory deterioration. Provide good supportive care, including intubation and ventilation, if necessary. Avoid opioids for pain management as they may mask symptoms of impending neurologic manifestations. Patients with suspected coral snake envenomations should be monitored for 12 hours after the initial bite [2].
Expert Commentary
Thank you, Dr. Lima for bringing the important and timely topic of snakebites to the table by posting this excellent overview! Current poison center data (2018 National Poison Data System) indicate a total of 4,013 crotalid exposures with the majority being copperheads. While morbidity is worrisome, mortality was fortunately low in our country with only one fatality reportedly from a rattlesnake [1].
Prehospital snakebite management has been an area of deserved scrutiny. Limb immobilization, analgesia, and transport to a medical facility are critical actions. Tourniquets, pressure immobilization bandages, cryotherapy, electrotherapy, and incision/suction are not recommended and are likely harmful. One researcher discovered that venom extraction suction devices “just suck” [2]. Having a cell phone in the field is most important to prevent loss of limb or life!
In other regions of the world, capturing or killing the snake may be optimal in determining which species specific antivenom to administer. For North American crotalids, however, this practice is discouraged and exceedingly dangerous. Both CroFab and Anavip (recently approved and now marketed with the goal of reducing risks of late coagulopathy) are prepared from several species of North American crotalids and can be used to manage any crotalid envenomation. These contemporary antivenoms (Fab fragments) are safer than older polyvalent antivenom that resulted in high rates of anaphylaxis.
Consult your regional poison center (1-800-222-1222) or staff medical toxicologist when managing snakebites! For the number of snakebites that present to the emergency department, poison centers manage severalfold more each year. Making decisions regarding the management of a limb that resembles compartment syndrome (more antivenom vs. surgical consultation), the interpretation of laboratory results, redosing of antivenom to gain initial control of swelling, and the management of nonindigenous (e.g. cobras, gaboon vibers) pet snakebites are nuances your subspecialists would love to collaborate on!
References
1. Gummin DD, Mowry JB, Spyker DA, BrooksDE, Beuhler MC, RiversLJ, Hashem HA, & Ryan ML 2018 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 36th Annual Report, Clinical Toxicology, 2019;57:1220-1413.
2. Bush SP. Snakebite Suction Devices Don’t Remove Venom: They Just Suck. Annals of Emergency Medicine, 2004;43:187-188.
Sean Bryant, MD
Assistant Director, Toxicology Fellowship Program, Department of Emergency Medicine, Cook County Health
Associate Professor, Department of Emergency Medicine, Rush Medical College
How To Cite This Post:
[Peer-Reviewed, Web Publication] Lima, R. Cornardy, M. (2020, Oct 26). Management of Snake Bite Injuries. [NUEM Blog. Expert Commentary by Bryant, S]. Retrieved from http://www.nuemblog.com/blog/snake-bites.
Other Posts You May Enjoy
References
Snakebite Injuries Treated in United States Emergency Departments, 2001–2004. O’Neil, Mary Elizabeth et al. Wilderness & Environmental Medicine, Volume 18, Issue 4, 281 - 287
Gold, Barry S., et al. “Bites of Venomous Snakes.” New England Journal of Medicine, vol. 347, no. 5, 1 Aug. 2002, pp. 347–356., doi:10.1056/nejmra013477.
Seifert, Steven A., et al. “AAPCC Database Characterization of Native U.S. Venomous Snake Exposures, 2001–2005.” Clinical Toxicology, vol. 47, no. 4, 2009, pp. 327–335., doi:10.1080/15563650902870277.
“Venomous snakes distribution and species risk categories.” World Health Organization. 2010. http://apps.who.int/bloodproducts/snakeantivenoms/database/
Cardwell, Michael D. “Recognizing Dangerous Snakes in the United States and Canada: A Novel 3-Step Identification Method.” Wilderness & Environmental Medicine, vol. 22, no. 4, 1 Oct. 2011, pp. 304–308., doi:10.1016/j.wem.2011.07.001.
Prophylactic Antibiotics Are Not Needed Following Rattlesnake Bites. August, Jessica A. et al. The American Journal of Medicine, Volume 131, Issue 11, 1367 - 1371
Recurrence phenomena after immunoglobulin therapy for snake envenomations: Part 2. Guidelines for clinical management with crotaline Fab antivenom. Annals of Emergency Medicine, 2001, Vol.37(2), p.196-201., doi: 10.1067/mem.2001.113134
Hall, Edward L. “Role of Surgical Intervention in the Management of Crotaline Snake Envenomation.” Annals of Emergency Medicine, vol. 37, no. 2, Feb. 2001, pp. 175–180., doi:10.1067/mem.2001.113373.
add tags
schedule post
share to twitter u%
format other posts you may enjoy
fix URL and copy to google file
U Tox: Clinical Utility and False Negatives
Written by: Ben Kiesel , MD (NUEM ‘23) Edited by: Jason Chodakowski, MD (NUEM ‘19) Expert Commentary by: Joe Kennedy, MD
Expert Commentary
The urine drug screen is one of the most frequently ordered and more frequently cursed tests in all of medicine. It is one of the few tests that generates more argument than the banal discussions between surgeons and emergency physicians regarding the utility of a white blood cell count. Nevertheless, in skilled hands, this cheap and simple immunoassay can answer a few quick questions. The key is in knowing your Achilles heel:
Opioids and opiates produce similar toxidromes, but there are virtually limitless combinations of street and prescription drugs, lab assays, and confirmatory tests to tell these apart. If the test is negative, who cares! Treat the patient in front of you. Fentanyl and its analogues are perhaps most often missed. Wake the patient up, discuss their substance use, and move on.
Hopefully you noticed that *many* things other than phencyclidine can lead to a positive urine drug screen for PCP. Ever have a conversation with the mother of a 12 year old about their PCP use? Best go into that conversation knowing that many other substances cross-react. Perhaps the kid is actually on lamotrigine for seizures, and that is why they were combative. Or perhaps like most humans, they took a few ibuprofen at some point. Either way—when child protective services is consulted for a positive result, either call the poison center or your local toxicologist for help.
Perhaps the most important point is that this is a screen and absolutely by no means a confirmatory test. When it matters (placement, custody, fired/hired, transplant eligibility, excluding other diagnoses), get an actual level or result. Not sure how to do that? Call your lab! Or call me. Toxicologists love solving these problems and teaching and helping others do the same!
Joe Kennedy, MD
Attending Physician, Emergency Medicine
University of Illinois Hospital
Senior Toxicology Fellow
Toxikon Consortium
How To Cite This Post:
[Peer-Reviewed, Web Publication] Kiesel, B. Chodakowski, J. (2020, Oct 19). U Tox: Clinical Utility and False Negatives. [NUEM Blog. Expert Commentary by Kennedy, J]. Retrieved from http://www.nuemblog.com/blog/utox-clinical-utility-and-false-negatives.
Other Posts You May Enjoy
Vaporizing Lung Injury
Written by: Aaron Wibberley, MD (NUEM ‘22) Edited by: Matt McCauley, MD (NUEM ‘21) Expert Commentary by: Leon Gussow, MD
Initial post
Expert Commentary
Although the large cluster of EVALI cases seen last summer and fall has subsided, the known and potential pulmonary problems associated with vaping nicotine or THC products remain an important topic for emergency practitioners and medical toxicologists alike. In a recent update on EVALI, the CDC reported that as of February 18, 2020 a total of 2807 cases had been documented from all 50 states, the District of Columbia, Puerto Rico, and the U.S. Virgin Islands. Among these cases were 68 fatalities. [1]
As this instructive post by Drs. Wibberley and McCauley suggests, many vaping liquids available at retail outlets or on the street are largely unregulated and may contain a witch’s brew of additives and contaminants whose effects on the human respiratory system have not been adequately studied. In addition to glycerin, propylene glycol, and various flavorings, inhaled vapor from these products may also contain toxic metals, formaldehyde, nitrosamines, and acrolein. [2]
One additive strongly linked to EVALI is vitamin E acetate, a synthetic oil used commercially in skin creams, dietary supplements, and multivitamins. Vitamin E acetate has been detected in many non-commercial illicit THC vaping cartridges used by EVALI patients, where it might have been added as a thickener. It was also found in bronchoalveolar lavage (BAL) fluid drawn from 48 of 51 (94%) confirmed or probable cases of EVALI, but in no such samples from 99 healthy controls. [3,4] Vitamin E acetate may impair the function of pulmonary surfactant. Despite this strong link, the CDC concluded that “evidence is not sufficient to rule out the contribution of other chemicals of concern.’ [5]
As noted in the post, since EVALI is a diagnosis of exclusion, initial clinical efforts should focus on supportive care and ruling-out other potential causes, especially pulmonary infections. Suspecting the diagnosis and establishing a connection to vaping is particularly challenging during flu season or large outbreaks of other respiratory infections. But if EVALI is not considered, a relatively stable patient with early disease may be sent home only to resume vaping. That could lead to disaster. Although new cases of EVALI have not been reported in the last several months, here’s what I think is good practice: any patient with new respiratory complaints should be asked about vaping. If they partake, they should be advised that the practice may be exacerbating their symptoms and counseled to abstain.
References
Outbreak of Lung Injury Associated with E-cigarette Use, or Vaping. Centers for Disease Control and Prevention. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html#latest-information. Accessed May 10, 2020.
Ind PW. E-cigarette or vaping product use-associated lung injury. Br J Hosp Med. 2020 Apr;81(4):1-9.
Sun LH. Contaminant found in marijuana vaping products linked to deadly lung illnesses, tests show. Washington Post Sept 6, 2019.
Blount BC et al. Vitamin E Acetate in Bronchoalveolar-Lavage Fluid Associated with EVALI. N Engl J Med 2020;382:697-705.
Ghinai I et al. Characteristics of Persons Who Report Using Only Nicotine-Containing Products Among Interviewed Patients with E-cigarette, or Vaping, Product Use-Associated Lung Injury — Illinois, August-December 2019. MMWR 2020 Jan 24;69(3):84-89.
Dr. Leon Gussow, MD
Assistant Professor of Emergency Medicine, Rush University
Consultant for Illinois Poison Center
Medical Editor, The Poison Review
How To Cite This Post:
[Peer-Reviewed, Web Publication] Wibberley, A. McCauley, M. (2020, Sept 28). Vaporizing Lung Injury. [NUEM Blog. Expert Commentary by Gussow, L]. Retrieved from http://www.nuemblog.com/blog/vaporizing-lung-injury
Other Posts You May Enjoy
Lipid Emulsion Therapy for Local Anesthetic Systemic Toxicity
Written by: Dana Loke, MD (NUEM ‘20) Edited by: Jim Kenny, MD (NUEM ‘18) Expert Commentary by: Patrick Lank, MD, MS
Local anesthetic systemic toxicity (LAST) is a feared complication of local anesthetic use. Current estimates of LAST toxicity in adults range from 7.5 to 20 per 10,000 peripheral nerve blocks and 4 per 10,000 epidurals.[1] Although rare, this complication can be fatal. Unfortunately, many physicians are unaware of the toxic dose of local anesthetics and are unable to recognize the signs and symptoms of this toxicity.[2] For this reason and the fact that local anesthetic toxicity is rare, by the time this syndrome is identified, patients are often in cardiac arrest or peri-arrest. Thankfully, lipid emulsion such as Intralipid is a safe and effective therapy used to treat LAST.
How does lipid emulsion therapy work?
Lipid emulsion therapy is an intravenous therapy that binds lipophilic toxins and therefore reverses their toxicity. There are several brand name lipid emulsion therapies, however Intralipid, a soy-based lipid emulsion that contains long-chain triglycerides, is the most commonly used (Figure 1).[3] The ability of lipid emulsion therapy to counteract the toxic effects of local anesthetics was discovered in 1998 by Weinberg et al when it was incidentally found that lab rats pre-treated with an infusion of lipids could withstand larger doses of bupivacaine before arresting.[4] The rats were also more easily resuscitated if given lipid emulsion therapy.[1] These findings were subsequently confirmed in other laboratories and clinical systemic analyses.[5] Once studied more directly, it was found that intralipid acts as a “sink” by creating a lipid compartment within the plasma that attracts lipophilic compounds, such as local anesthetics, into the lipid sink, which is separate from the aqueous phase of the plasma.[1]
Figure 1: Composition of Various Brands of Lipid Emulsions[1]
How does LAST manifest?
Toxicity is a rare but potentially lethal side effect of local anesthetic. However, since patients often present without any knowledge that they were administered toxic doses of local anesthetic, it is important that the EM physician be cognizant of the signs of this toxicity. Symptoms typically start after a toxic dose of local anesthetic is administered or if local anesthetic is inadvertently administered directly into a vessel instead of subcutaneously (Figure 2). Onset of LAST is typically 30 seconds to 60 minutes after administration of the anesthetic but more often than not occurs within 1-5 minutes.[6]
Figure 2: Maximum Doses and Durations of Various Local Anesthetics[9]
Symptoms of LAST can vary, however there are 5 general ways in which LAST presents.[6] One or all of these manifestations may be present.
CNS (excitement) – an early manifestation of LAST that often begins with confusion or slurred speech but may include subjective symptoms like metallic taste in the mouth, tinnitus, oral numbness, dizziness, lightheadedness, or visual or auditory disturbances. If not treated promptly, these symptoms often progress to seizures, syncope, coma, respiratory depression, or cardiovascular collapse.
Cardiovascular – often preceded by CNS symptoms but not always. May include hypertension, tachycardia or bradycardia, arrhythmias, and asystole. Depressed contractility of the heart then leads to progressive hypotension and ultimately cardiac arrest.
Hematologic – methemoglobinemia, cyanosis
Allergic – urticaria, rash, and rarely anaphylaxis
Local tissue response – numbness, paresthesia
The EM physician should maintain a high level of suspicion should a patient present after a same day surgery or procedure with any constellation of these symptoms.
How is lipid emulsion therapy administered?
Once LAST is recognized, the EM physician should immediately consider giving lipid emulsion therapy. An initial dose of 20% lipid emulsion at 1.5 ml/kg or a 100 ml bolus can be administered over a few minutes. This can be repeated after 5 minutes for 2 or more times for persistent hemodynamic instability. The bolus(es) should immediately be followed by a continuous infusion at 0.25-0.5 ml/kg/min.[3] The infusion should run for a minimum of 10 minutes after return of hemodynamic stability, however there are documented reports of recurrent systemic toxicity even after this. For this reason, patients should be admitted for at least 12 hours for observation and additional doses of intralipid as needed for rebound symptoms or hemodynamic compromise.[3] Consultation with your facility’s poison center is also crucial to further guide management.
Efficacy
In terms of efficacy, case reports and systemic analyses have found that lipid emulsion therapy:
Can reverse both neurologic and cardiac toxicity [5]
Leads to significantly higher rates of ROSC compared to saline controls in animal models [5]
Is more effective for witnessed events (for example, brief down time for patients that arrest)5
Is often effective in patients in which epinephrine, vasopressin, and antiarrhythmic medications did not work
Both hypoxia and acidosis worsen the toxicity of local anesthetics and may inhibit lipid emulsion therapy, so it is imperative that oxygenation and acid-base status are optimized when lipid emulsion therapy is needed.[3, 5]
Contraindications, Complications, and Special Populations
There are no absolute contraindications to intravenous lipid emulsion therapy and no clinically significant complications documented in the literature. The benefits of lipid emulsion therapy will often outweigh any potential risks in patients with LAST, especially if hemodynamically unstable or coding.
Potential complications of lipid emulsion therapy are mainly related to hypersensitivity. Patients allergic to soybean protein or eggs theoretically may develop allergic or anaphylactic reactions. These reactions should be treated like all other allergic or anaphylactic reactions: with anti-histamines, steroids, and epinephrine as needed. Additionally, there are reported cases of hyperamylasemia however no documented progression to clinical pancreatitis.[3] There are also case reports of extreme lipemia, however even a patient that was inadvertently given 2 L of 20% lipid emulsion did not develop any cardiopulmonary complications.[5] The lipemia however did interfere with standard laboratory tests.[5]
Intralipid is safe in pregnancy and has documented use for treating LAST in term pregnancy.[7] Furthermore, it has documented uncomplicated use in pediatric and neonatal patients.[3, 8]
Key Points
Systemic toxicity is a rare but potentially fatal complication of local anesthetic use.
Lipid emulsion therapy such as Intralipid mitigates the toxic effects of local anesthetics and can reverse both neurologic and cardiac toxicity.
LAST may manifest initially with CNS symptoms but can progress to seizure, respiratory depression, coma, and cardiovascular collapse.
An initial bolus of 1.5 ml/kg or 100 ml 20% lipid emulsion followed by an infusion starting at 0.25 ml/kg/min is crucial to reverse toxicity and prevent recurrence.
Hypoxia and acidosis both worsen LAST and may inhibit lipid emulsion therapy.
Patients should be admitted in order to monitor for recurrent toxicity.
There are no contraindications to and minimal side effects of lipid emulsion therapy.
Expert Commentary
Thank you both for the above thorough review of local anesthetic systemic toxicity (LAST) from the emergency physician perspective! I only want to add a few points to consider when learning more about LAST.
Without going into too much detail, there has been a lot of research done to figure out exactly how lipids aide in the treatment of patients with severe LAST. The lipid sink model is wonderfully understandable and explains many of the clinical and laboratory we see (e.g., a greater decrease in free serum concentration of more lipophilic local anesthetics). However, there are some other models and theories to be aware of. One I am fascinated by is the “lipid shuttle.” Fundamentally, this describes the phenomenon that lipid therapy will decrease the concentration of local anesthetic at sites of toxicity (i.e., heart and CNS) and increase its concentration in the liver. So instead of lipids acting only as a “sink” to remove a toxin from free availability, it is helping mobilize the toxin to an area where it can go through the process of elimination from the body. Additionally, there are wonderful biochemical explanations (e.g., fatty acid supply, inhibition of nitric oxide release, reversal of mitochondrial dysfunction) to the positive cardiovascular effects seen after lipid treatment in LAST. All of these explanations, it seems, combine to contribute to the hemodynamic response seen in LAST.
Second, I would like to point your readers towards a resource that may help them work through the mechanics of administering lipid rescue therapy in LAST – lipidrescue.org. On that website, one can find links to various protocols, compilations of prior research done on the topic, and much more background on the science of the treatment than I provided above.
Third, in the emergency department, I think you are correct in saying that the most likely source of LAST we would see would come from outpatient surgery centers. A few other clinical scenarios to be aware of would include the following: ingestion of local anesthetics – mostly benzonatate (Tessalon); non-surgical outpatient aesthetic offices that may use topical anesthetics; inappropriate and excessive home use of local anesthetics for pain relief.
Finally, a very brief comment on the use of lipid rescue therapy in non-LAST toxic exposures although that was not the subject of your post. While lipid rescue therapy for LAST has a remarkable record of being effective, that is not yet the case with its use in other toxic exposures. A list of the side effects of lipid rescue therapy includes but is not limited to ARDS, pancreatitis, infection, and significant laboratory interference. While in the setting of severe LAST, the risk: benefit often favors administering lipid rescue, this may not be the case in the setting of non-LAST exposures. For those non-LAST cases (as well as with LAST cases) in which you are wondering if lipid rescue would be appropriate, I would strongly recommend you call your regional poison center to discuss further focused therapy.
Patrick Lank, MD, MS
Assistant Professor of Emergency Medicine
Medical Toxicologist
Department of Emergency Medicine
How To Cite This Post:
[Peer-Reviewed, Web Publication] Loke D, Kenny J. (2020, July 20). Lipid Emulsion Therapy for Local Anesthetic Systemic Toxicity. Expert Commentary by Lank P. Retrieved from http://www.nuemblog.com/blog/lipid-emulsion-therapy
Other Posts You May Enjoy
References
1. Manavi, M. (201). Lipid infusion as a treatment for local anesthetic toxicity: a literature review. AANA Journal, 78(1), 69-78.
2. Cooper, B.R., Moll, T., & Griffiths, J.R. (2010) Local anaesthetic toxicity: are we prepared for the consequences in the Emergency Department. J Emerg Med, 27(8), 599.
3. Mercado, P. & Weinberg, G.L. (2011). Local anesthetic systemic toxicity: prevention and treatment. Anesthesiology Clin, 29(2), 233-242.
4. Weinberg, G.L., VadeBancouer, T., Ramarju, G.A., Garcia-Amaro, M.F., & Cwik, M.J. (1998). Pretreatment or resuscitation with a lipid infusion shifts the dose-response to bupivacaine-induced asystole in rats. Anesthesiology, 88(4), 1071-5.
5. Weinberg, G.L. (2012). Lipid emulsion infusion: resuscitation for local anesthetic and other drug overdose. Anesthesiology, 117(1), 180-7.
6. Wadlund, D. (2017). Local anesthetic systemic toxicity. ARON Journal, 106(5), 367-77.
7. Dun-Chi Lin, J., Sivanesan, E., Horlocker, T.T., & Missair, A. (2017). Two for one: a case report of intravenous lipid emulsion to treat local anesthetic systemic toxicity in term pregnancy. A&A Case Reports, 8(9), 235-7.
8. Shah, S., Gopalakrishnan, S., Apuya, J., Shah, S., & Martin, T. (2009). Use of intralipid in an infant with impending cardiovascular collapse due to local anesthetic toxicity. J Anesth, 23(3), 439-441.
9. “Missouri Society of Health-System Pharmacists - Overview of Management of Local Anesthetic Systemic Toxicity (LAST) Based on Updated 2017/18 ASRA Practice Guidelines.”
Mood Stabilizer Toxicities
Written by: Justine Ko, MD (NUEM PGY-4) Edited by: Sarah Dhake, MD (NUEM ‘19) Expert Commentary by: Patrick Lank, MD, MS
Expert Commentary
This is a great summary of the causes, symptoms, work-up, and treatment of two relatively common medications that cause toxicity. In fact, these (along with carbamazepine) are levels I routinely recommend checking in patients with a history of bipolar disorder who come to the emergency department with altered mental status even if they do not report a history of being on these medications. That is because these three medications are very commonly used in the treatment of bipolar disorder and all have quite different treatment courses. In addition to the great summary above, below are some of my usual teaching points about these medications in overdose.
Let's tackle them separately as they are quite different toxicities.
First let's talk about lithium. In almost all medical texts, the tissue distribution of lithium is appropriately identified as being "complex." The easiest way I communicate that with patients, families, and medical learners is that in chronic therapy, lithium forms "stores" of drug in the body and intracellularly. Clinically that is relevant because after performing hemodialysis (HD) for lithium toxicity, you will reliably see an initial drop in lithium concentration followed by elevation approaching pre-dialysis levels if routine HD is performed. Although that could make one feel ambivalent about routinely recommending HD for lithium toxicity, there is suggestion of an alternate advantage of performing HD for lithium toxicity. Vodovar et al published a study in 2016 showing that patients who met their institutional criteria for HD and had HD performed had significantly fewer neurologic side effects from their toxicity than those who met criteria but did not have HD. So even though it did not impact usual measurements that we would expect HD to influence -- mortality and ICU length of stay -- its performance in this study seems to have been clinically beneficial.
The other big thing to discuss with lithium toxicity is that there are many known medication interactions with lithium. In short, any medications that impair renal function should not be used in someone on chronic lithium therapy. The main list of those medications includes NSAIDs, ACE inhibitors, ARBs, and thiazide diuretics.
Most of the unique aspects of valproate toxicity focus on its diagnosis and treatment. In the setting of acute intentional overdose of valproate, one of the most important things for emergency physicians to be aware of is that there can be a delay of peak valproic acid level. There are cases of patients presenting to an emergency department with stated valproate ingestion, initial negative level, then repeat level hours later being in the toxic range. So I recommend serial valproate levels until both down-trending and non-toxic. For treatment, there is a great summary of recommendations by the Extracorporeal Treatments in Poisoning Workgroup (EXTRIP) published online (https://www.extrip-workgroup.org/valproic-acid). In short, consult nephrology for dialysis if the patient is super sick.
As always, I recommend you consult your regional poison center when you are worried your patient is experiencing medication toxicity. But I hope this infographic and some of my comments helps you understand their recommendations.
References
Vodovar V, et al. Lithium poisoning in the intensive care unit: predictive factors of severity and indications for extracorporeal toxin removal to improve outcome. Clin Tox (Phila) 2016 Sep; 54(8): 615-23.
Lank P and Bryant S. "Valproic Acid" In: Wolfson A, Hendey G, Ling L, Rosen C, Schaider J, Cloutier R (eds.): Harwood-Nuss’ Clinical Practice of Emergency Medicine, 6th edition. Lippincott Williams & Wilkins 2014.
Patrick Lank, MD, MS
Assistant Professor of Emergency Medicine
Medical Toxicologist
Department of Emergency Medicine
How To Cite This Post
[Peer-Reviewed, Web Publication] Ko J, Dhake S. (2020, July 13). Mood Stabilizer Toxicities [NUEM Blog. Expert Commentary by Lank P. Retrieved from http://www.nuemblog.com/blog/mood-stabilizer-tox
Other Posts You May Enjoy
Alcohol Related ED Visits
Written by: Elizabeth Stulpin, MD (NUEM PGY-1) Edited by: Kevin Dyer, MD (NUEM PGY-2) Expert Commentary by: Erin Lareau, MD
Alcohol related ED visits are an all too common occurrence and are only becoming more frequent. Between 2006 and 2014, the number of ED visits involving alcohol consumption increased by over 60 percent. This has led us to our current estimated 1.2 million hospital admissions per year related to alcohol abuse, with about 500,000 of those requiring greater attention for acute withdrawal.
Alcohol withdrawal can be treated with an innumerable combination of medications and dosages, with symptom dosed benzodiazepines currently the treatment of choice. However, with increasing visits for withdrawal and increasing doses needed to control symptoms, adverse effects of benzodiazepines can also be seen, such as paradoxical agitation, propylene glycol toxicity from increasing doses of lorazepam (lactic acidosis, AKI) and respiratory depression requiring intubation. Some patients are also resistant to benzodiazepine therapy, showing no symptomatic relief despite increasing doses. These factors have led to a pendulum swing back to the age before benzodiazepines, when phenobarbital was a first line treatment for treating alcohol withdrawal.
Benefits of Phenobarbital:
Reaching for phenobarbital instead of the syringe or tablet of lorazepam, diazepam, or chlordiazepoxide is an attractive option for many reasons. From its mechanism to pharmacokinetics to side effects, phenobarbital as a monotherapy can take some guesswork out of treatment. At the same time, patient outcomes are shown to be similar or improved when compared to benzodiazepines.
Mechanism:
The underlying cause of alcohol withdrawal is multifactorial. With chronic alcohol use, inhibitory GABA receptors are down regulated, while excitatory glutamate and NMDA receptors are upregulated. When the inhibitory effect of alcohol is suddenly removed, an excessive excitatory state is produced, resulting in the typical symptoms of tremulousness, anxiety, tachycardia, among others. Like benzodiazepines, phenobarbital targets GABA receptors to decrease excitatory tone, but does so more effectively since it does not rely on the presence of endogenous GABA hormone. At the same time, phenobarbital also down regulates glutamate signaling, leading to a more comprehensive approach to treating withdrawal.
Pharmacokinetics:
Besides its mechanism of action (MOA), one of the most attractive features of phenobarbital therapy is its simple pharmacology. Not only can it be given IV, IM or PO with almost 100 percent bioavailability in each route, the drug level in the body is extremely predictable. A linear relationship exists between the cumulative weight based dose and the resulting plasma concentration, which allows providers to reliably achieve the target dose range while avoiding levels at which toxicity would occur. This is in contrast to benzodiazepines, where metabolism and clinical response vary greatly among patients, and drug levels are nearly impossible to predict when periodically dosing with different doses at nonstandard points of time.
Another added benefit of phenobarbital is its length of action. Whereas the half-life of lorazepam is 14 to 20 hours, phenobarbital has a half-life of 80 to 120 hours. This allows for titration of medication over the patient’s hospital stay and an auto-tapering effect that can prevent rebound symptoms for days after, one of the main benefits of using chlordiazepoxide.
Patient outcomes:
The superior molecular characteristics and MOA of phenobarbital carry over to patient outcomes as well. Since the 1970s, phenobarbital monotherapy has been shown to be as safe, and as or more effective than benzodiazepines. With renewed focus on phenobarbital, more recent studies have been conducted that corroborate this claim. In the past few years, phenobarbital has been shown to have similar or improved outcomes when compared to benzodiazepines in terms of ICU stay and overall length of hospital stay. Additionally, patients treated with phenobarbital monotherapy have been seen to have decreased rates of delirium, decreased need for additional PRN sedation and lower rates of leaving against medical advice.
With regards to the most feared outcome, respiratory depression, phenobarbital has also held its own. Patients treated with phenobarbital have shown to have similar or even reduced rates of intubation, possibly due to the predictable linear relationship between drug dosing and plasma concentration as discussed previously. Secondly, the therapeutic range of phenobarbital is quite large. While the therapeutic dose hovers around 5 to 25mg/kg of total body weight, the toxic dose requiring intubation is often greater than 40mg/kg.
Phenobarbital was also shown to be effective in patients whose symptoms were refractory to benzodiazepines, possibly due to its different mechanism and lack of dependence on endogenous GABA.
What now?
So now you want to give phenobarbital a try, what now?
The most commonly seen dosing regimen is a loading dose, followed by IV or PO titration to the patient’s therapeutic level based on symptoms.
The IV loading dose is often set at 10mg/kg ideal body weight, infused over 30 minutes to achieve a serum level of about 15ug/mL. Similar to treatment with benzodiazepines, treatment is then symptom based. Repeat infusions or pills of 130mg are given every 15 minutes, with a total upper limit of 20 to 30mg/kg. Unlike benzodiazepine therapy where steadily increasing doses are used to control agitation, phenobarbital’s standard dosing takes some of the guesswork out of treatment for providers. Once symptoms have been appropriately managed, the patient may require additional PRN doses of phenobarbital, but the slow auto-tapering effect of the drug should prevent acute decompensation.
Take Home Points:
Phenobarbital is a safe and effective method to combat the symptoms of alcohol withdrawal.
Start low and go slow! If the patient has received benzodiazepines already, phenobarbital will act synergistically, and high doses may increase the risk of respiratory depression.
Remain aware of the cumulative phenobarbital dose to prevent reaching levels at which toxicity would occur.
Talk to your pharmacist about using phenobarbital for your next case of alcohol withdrawal!
Expert Commentary
This is a wonderful review of current treatment strategies for alcohol withdrawal in the ED. To broaden and emphasize some of your main concepts:
Alcohol related ED visits are extremely common. ED patients with alcohol related chief complaints are high risk for traumatic, medical, psychiatric and toxicologic problems that can easily be missed if providers are not vigilant.
Some patients may present to the ED with chief complaints that are medical or psychiatric in nature, but are directly associated with an underlying alcohol use disorder, making their management even more challenging.
Patients with unhealthy alcohol use may present to the ED with intoxication, withdrawal, seizures, agitation/psychosis, falls, traumatic injuries, gastritis/ GI bleeding, liver disease, cardiac disease, depression and anxiety. Many have concurrent social challenges including domestic violence and homelessness.
When a patient with an alcohol use disorder presents in acute withdrawal, there are multiple treatment strategies to use for symptom relief. Benzodiazepines are widely used and effective, though often require repeat doses to obtain maximal effect. Phenobarbital loading is a wonderful alternative with similar outcomes and simplified, standard dosing, as noted by the pharmacokinetics you reviewed.
My personal clinical practice is to preferentially use phenobarbital for high risk patients who present for alcohol withdrawal symptoms, and who have not already received IV benzodiazepines. Those at high risk for complications from alcohol withdrawal may have a history of:
withdrawal seizures
hallucinations
delirium tremens
abuse of multiple substances
recent admission (ICU or floor) for severe alcohol withdrawal.Phenobarbital loading is also effective for patients who are not responding to high dose IV pushes of benzodiazepines, as an alternative to starting continuous infusions. Anecdotally, I have spared patients such as this from requiring intensive care admission for withdrawal by using this pathway, though I have not found any research to support that this is a widespread phenomenon. Because serum phenobarbital levels can be checked for maintenance of a therapeutic range, severe withdrawal requiring multiple repeat doses can be directed in a more simplified way. For those with more mild symptoms, PO or IV benzodiazepines continue to be appropriate and have good effect.
Be sure to screen your patients presenting with alcohol intoxication or withdrawal for other emergent medical conditions, including traumatic, medical and psychiatric. Similarly, think about alcohol abuse and withdrawal in patients presenting for other problems, but who exhibit abnormal vital signs, abnormal neurologic exam or an insufficient response to treatment. Ask yourself:
Are there signs of trauma?
Are there other intoxicants?
Does this medical patient have untreated alcohol withdrawal? Is there tremor, hypertension and tachycardia that is not otherwise explained?
Are there vital sign abnormalities in the intoxicated patient? Can they be explained by intoxication? By withdrawal? If the answer is no, be sure you have appropriately treated for dehydration, alcoholic ketoacidosis and/or alcohol withdrawal. Then look for concomitant disease.
Was this patient trying to hurt themselves? Do they have underlying psychiatric disease? Are they in treatment? Do they need further resources?
Even a single conversation may be a life changing intervention for someone with an underlying alcohol use disorder!
References:
UpToDate article on “Risky Drinking and Alcohol Use Disorder” https://www.uptodate.com/contents/risky-drinking-and-alcohol-use-disorder-epidemiology-pathogenesis-clinical-manifestations-course-assessment-and-diagnosis?search=risky-drinking-and-alcohol-use-&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1. Accessed on 2/09/2020.
Hammond, D, et al. “Patient Outcomes Associated With Phenobarbital Use With or Without Benzodiazepines for Alcohol Withdrawal Syndrome: A Systematic Review.” Hospital Pharmacy 2017;52(9):607-616
Erin Lareau
Assistant Professor
Department of Emergency Medicine
Feinberg School of Medicine
Northwestern University
How to Cite This Post
[Peer-Reviewed, Web Publication] Stulpin, E, Dyer, K. (2020, May 25). Alcohol Related ED Visits [NUEM Blog. Expert Commentary by Lareau, E]. Retrieved from https://www.nuemblog.com/blog/etoh
References
Farkas, Josh. “Alcohol Withdrawal.” The Internet Book of Critical Care. November, 2016. http://www.emcrit.org/ibcc/etoh/.
Gortney, J, et. al. “Alcohol withdrawal syndrome in medical patients.” Cleveland Clinic Journal of Medicine 2016; 83(1): 67-79.
Hammond, D, et. al. “Patient Outcomes Associated With Phenobarbital Use With or Without Benzodiazepines for Alcohol Withdrawal Syndrome: A Systematic Review.” Hospital Pharmacy 2017; 52(9): 607-616.
Hendey, G, et. al. “A prospective, randomized, trial of phenobarbital versus benzodiazepines for acute alcohol withdrawal.” The American Journal of Emergency Medicine 2011; 29(4): 382-385.
Hsu, D, et. al. “Phenobarbital versus Benzodiazepines for the Treatment of Alcohol Withdrawal Syndrome in Critically Ill Patients.” American Journal of Respiratory and Critical Care Medicine 2015; 191(A3704):
Kattimani, S and Bharadwaj, B. “Clinical management of alcohol withdrawal: A systematic review.” Industrial Psychiatry 2013; 22(2): 100-108.
Kramp, P and Rafaelsen, OJ. “Delirium tremens: a double-blind comparison of diazepam and barbital treatment.” Acta Psychiatrica Scandinavica 1978; 58(2): 174-190.
Nelson, A, et. al. “Benzodiazepines vs barbituates for alcohol withdrawal: Analysis of 3 different treatment protocols.” The American Journal of Emergency Medicine 2019; 37(4): 733-736.
Nisavic, M, et. al. “Use of Phenobarbital in Alcohol Withdrawal Management – A Retrospective Comparison Study of Phenobarbital and Benzodiazepines for Acute Alcohol Withdrawal Management in General Medical Patients.” Psychosomatics 2019; 60(5); 458-467.
Rosenson, J, et. al. “Phenobarbital for acute alcohol withdrawal: a prospective randomized double-blind placebo-controlled study.” The Journal of Emergency Medicine 2013; 44(3): 592-598.
Tidwell, W, et. al. “Treatment of Alcohol Withdrawal Syndrome: Phenobarbital vs CIWA-AR Protocol.” American Journal of Critical Care 2018; 27(6): 454-460.
White, A, et. al. “Trends in Alcohol-Related Emergency Department Visits in the United States: Results from the Nationwide Emergency Department Sample, 2006 to 2014.” Alcoholism: Clinical and Experimental Research 2018; 42(2): 352-359.
Serotonin Syndrome
Written by: Jason Chodakowski, MD (NUEM PGY-3) Edited by: Evan Davis, MD (NUEM Alum ‘18) Expert commentary by: Benjamin Schnapp, MD (NUEM Alum ‘16)
Background
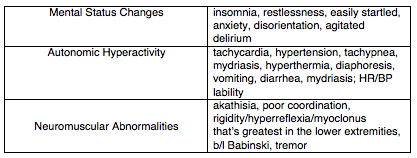
Serotonin syndrome is a condition characterized by increased serotonergic activity in the central nervous system. This can result from therapeutic use, inadvertent interactions, or intentional self-poisoning of any combination of drugs that have the net effect of increasing serotonergic neurotransmission. Serotonin syndrome most often causes mental status changes, autonomic and neuromuscular hyperactivity while severe cases may result in DIC, rhabdomyolysis, metabolic acidosis, renal failure, ARDS, and death.
Selective serotonin reuptake inhibitors (SSRIs) are the most commonly implicated group of medications, with other culprits including serotonin-norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs), and recreational drugs like cocaine, ecstacy, and amphetamines. The syndrome classically occurs after the initiation of a single drug, after increasing the dose of an existing medication, or simultaneous administration of two serotonergic agents. Serotonin syndrome involving a monoamine oxidase inhibitor may be especially severe and more likely to result in death. It’s important to keep in mind that antidepressants aren’t the only serotonergic agents, and that many commonly administered medications such as tramadol, meperidine, fentanyl, dextromethorphan, and linezolid among others have serotonergic activity.
Clinical Features
The majority of cases present within 24 hours, and most within 6 hours of a change or initiation of drug. The syndrome is characterized by the triad of mental status changes, autonomic hyperactivity, and neuromuscular abnormalities. The diagnosis, however, can be quite challenging as these three non-specific domains of symptomatology can present with very wide spectrum of severity.
Diagnosis
There is no confirmatory test for serotonin syndrome, the diagnosis is entirely clinical. The best validated diagnostic criteria for serotonin syndrome is the Hunter Toxicity Criteria. To fulfill the Hunter Criteria, a patient must have taken a serotonergic agent and meet one of the following conditions:
Spontaneous clonus
Inducible clonus plus agitation or diaphoresis
Ocular clonus plus agitation or diaphoresis
Tremor plus hyperreflexia
Hypertonia plus temperature above 38ºC plus ocular clonus or inducible clonus
Differential Diagnosis:
Multiple other entities can present with a general sympathomimetic picture and should be considered in the differential diagnosis. These include:
Neuroleptic malignant syndrome (NMS)
Anticholinergic toxicity
Malignant hyperthermia
Intoxication from other sympathomimetic agents
Opioid withdrawal
Sepsis
Meningitis/encephalitis
Heat stroke
Delirium tremens
Thyroid storm
Neuromuscular findings, particularly myoclonus is an important distinguishing feature of serotonin syndrome from the above etiologies.
Serotonin syndrome can typically be differentiated from other similar conditions by its characteristic neuromuscular findings of hyperreflexia and clonus. For example, conditions such as NMS, malignant hyperthermia, and sympathomimetic toxicity all typically lack these features.
Labs/Imaging
While serotonin syndrome is a clinical diagnosis labs and imaging can be useful for monitoring complications in severe cases and to help differentiate from other diagnoses in the differential above. The most severe complications of serotonin syndrome include DIC, rhabdomyolysis, metabolic acidosis, renal failure, and ARDS. Thus, you should consider checking the following to evaluate for these:
CBC
BMP
CK
LFTs
Lactic acid
DIC panel
UA
CXR
If the diagnosis is uncertain, you might also consider blood cultures, CT brain, lumbar puncture, urine toxicology screen and/or TSH to evaluate for other things on the differential.
Management
The mainstay of therapy for serotonin syndrome is supportive care. The main hallmarks of management include:
Discontinue the offending agent.
IV fluids to correct hypovolemia
Sedation with benzodiazepines. Options include Lorazepam 2-4mg and Diazepam 5-10mg, which can be repeated every 10 minutes.
Aggressive control of hyperthermia with standard cooling techniques. Since hyperthermia is often due to increased muscle activity in serotonin syndrome, consider early intubation and paralysis in severe hyperthermia.
Management of autonomic instability. Consider esmolol and nitroprusside for hypertension and tachycardia, while avoiding long-acting agent like propranolol. MAOIs can sometimes cause hypotension, treat this with pressors such as phenylephrine, epinephrine, and norepinephrine as necessary.
If benzodiazepines and supportive care fail to improve agitation and correct vital signs, you can consider cyproheptadine, an antidote of sorts with anti-histaminergic and anti-serotonergic activity. Cyproheptadine is only dosed orally, with a recommended initial dose of 12mg, followed by 2 mg every two hours until clinical response is seen. Symptoms from serotonin syndrome typically resolve within 24 hours.
Expert Commentary
Gathering an accurate medication history is the key to making this diagnosis. As mentioned above, this is a clinical entity that generally manifests quite quickly after medication initiation. Checking the medication list in the computer isn’t going to help you here - ask the patient what kinds of new medication they might have been prescribed or they might be taking. Remember - selective serotonin reuptake inhibitors (SSRIs) are not the only class of medications that can affect serotonin and it may be the interacting agent that you’re looking for. Besides the above, other common medications that can be problematic include triptans, ondansetron, valproic acid, carbamazepine, lithium, cyclobenzaprine, and even herbal supplements like St. John’s wort (patients may not think supplements are worth mentioning!).
One underappreciated aspect of serotonin syndrome is the extent to which it occurs on a spectrum. Studying for the boards and looking at the Hunter Criteria can leave one with the impression that clonus is necessary to make the diagnosis of serotonin syndrome. This is not the case, and it has been suggested that many milder cases, presenting with only symptoms such as tachycardia, diarrhea, and restlessness, may be going missed. Again, medication history is essential here.
Benjamin Schnapp, MD
Assistant Residency Program Director, University of Wisconsin-Madison Emergency Medicine
How To Cite This Post
[Peer-Reviewed, Web Publication] Chodakowski J, Davis E (2018, November 19). Serotonin Syndrome [NUEM Blog. Expert Commentary by Schnapp B]. Retrieved from http://www.nuemblog.com/blog/serotonin-syndrome
Other Posts You May Enjoy
References
Boyer, Edward W., and Michael Shannon. "The serotonin syndrome." New England Journal of Medicine 352.11 (2005): 1112-1120.
LoVecchio, Frank, and Erik Mattison.. "Atypical and Serotonergic Antidepressants." Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8e Eds. Judith E. Tintinalli, et al. New York, NY: McGraw-Hill, 2016,
Mason, Peter J., Victor A. Morris, and Thomas J. Balcezak. "Serotonin Syndrome Presentation of 2 Cases and Review of the Literature." Medicine 79.4 (2000): 201-209.
Toxic Flames
Written by: Vidya Eswaran, MD (NUEM PGY-3) Edited by: Jonathan Andereck, MD (NUEM PGY-4) Expert commentary by: Matt Zuckerman, MD (University of Colorado)
Expert Commentary
Thank you, this highlights an important aspect of treating victims of smoke inhalation.
In terms of the physiology of CO I like to think of it as an acquired hemoglobinopathy at low doses, thus patients with premorbid cardiopulmonary disease may be affected at lower doses. A fair amount has been written about how absolute levels correlate poorly with clinical effects. The idea of levels correlating to symptoms seem to originate from a Bureau of Mines publication from 1923 that won’t disappear. I would suggest having a low threshold for testing anyone who might have exposure; the failure for CO is in not testing.
Additionally, cherry lips are rarely found in living patients (more commonly on autopsy event at levels below 50%) so are rarely clinically useful (J Forensic Sci. 1995 Jul;40(4):596-8).
The “consider” HBO recommendation for COHb levels >25% is very controversial and the literature is limited by heterogeneity in patients and treatment protocols. Some would argue against hyperbaric for most patients or even consider HBO for patients at lower levels. Consultation with toxicologists and hyperbaricists is likely to be helpful.
Lactic acidosis is key to cyanide poisoning. Most use a combination of smoke exposure with an elevated lactate (>10 mmol/L) to be highly suggestive of CN toxicity and an indication for empiric treatment. CN levels are rarely helpful and rarely ordered. The description of cyanide symptoms “progressing” is a bit of a misnomer as cyanide is initially rapid onset, without evolving symptoms; indeed knockdown is a common presenting symptom. Hydroxocobalamin is preferred to the antidote kit, and amyl nitrate is omitted if sodium nitrite is given. The transient hypertension associated with hydroxocobalamin is often therapeutic given the incidence of hypotension, and its important to be aware that this will discolor serum and tears and urine.
Matthew Zuckerman, MD
Assistant Professor of Emergency Medicine, University of Colorado School of Medicine
How to cite this post
[Peer-Reviewed, Web Publication] Eswaran V, Andereck J (2018, August 20). Toxic Flames. [NUEM Blog. Expert Commentary by Zuckerman M]. Retrieved from http://www.nuemblog.com/blog/toxic-flames
Posts you may also enjoy
Serotonin Syndrome
Written by: Jacob Stelter, MD (NUEM PGY-3) Edited by: Elizabeth Byrne, MD, (NUEM Graduate 2017) Expert Commentary by: Patrick Lank, MD
The Case
A 21-yearold male with history of PTSD and depression presented to the ED complaining of “feeling anxious.” When evaluated, the patient was found to be anxious, continuously pacing around the room. His speech was pressured as he tangentially answered questions. During the interview he stated that he was discharged a few days ago from a psychiatric hospital after he had intentionally overdosed on clonazepam. He then admited that two hours prior to arrival in your ED he took 30 pills of his prescribed sertraline 100mg tabs to “calm his nerves.” He denied any concurrent substance abuse. Despite the stated overdose, he denied any suicidal intent with his ingestion.
Physical Exam:
Vital Signs: T 98.9F oral HR 137 BP 152/89 RR 20 Sat 99% on room air
General: Awake, alert, anxious-appearing, pacing around the room, mildly diaphoretic
Head: normocephalic, atraumatic
HEENT: PERRL, EOMI, anicteric sclera, mydriatic pupils b/l, 8mm
Cardiac: tachycardic, regular rhythm, no MRG
Resp: clear to auscultation, nonlabored, no crackles appreciated
Abd: Soft, not distended, not-tender to palpation, +bowel sounds
Extremities: well perfused, 2+ radial and DP pulses b/l, mildly diaphoretic
Neuro: AAOx3, 5/5 strength in all 4 extremities, +ankle clonus, hyperreflexia on patellar reflexes.
Psych: Cooperative but anxious with pressured and tangential speech., no HI , SI, hallucinations/delusions
Pertinent Labs:
WBC 12.5, Hgb 15.8, electrolytes within normal limits, LA of 1.7.
Urine drug screen was positive for amphetamines and benzodiazepines. Acetaminophen, salicylate, and ethanol concentrations were below detection limits.
ECG:
Sinus tachycardia, QTc 457, no abnormal morphology of the QRS complexes
Diagnosis?
Serotonin Syndrome due to SSRI overdose.
Serotonin Syndrome
Overview
Serotonin syndrome is a disorder that is precipitated by excess serotonin. It is classically described as having a combination of hyperautonomic hemodynamic changes, neuromuscular derangements, and a change in the patient’s mental status. [1] Serotonin syndrome is a medical condition seen only in patients with exposure to serotonergic medications and has been more recently appreciated since the advent of serotonergic drugs used to treat depression and anxiety. However, in addition to pharmaceuticals, botanicals and recreational drugs of abuse can also be serotonergic. Causative substances increase the amount of circulating serotonin in the bloodstream, which leads to an increased propensity to develop serotonin syndrome [2]. This is not a rare diagnosis; there are approximately 7,300 cases per year results in about 100 deaths [1].
Clinical Presentation
Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med 2005;352:1112-20.
The diagnosis of serotonin syndrome will often manifest around the time of a dose increase, addition of another serotonergic agent or an overdose on a serotonergic agent [3]. Serotonin syndrome will typically manifest within 24 hours of exposure to the serotonergic agent [4]. Symptom presentation can be separated into three main categories: cognitive, autonomic and neuromuscular. Cognitive symptoms range from restlessness and anxiety to agitation and altered mental status [2]. Autonomic signs and symptoms include tachycardia, mydriasis, hyper- or hypotension, hyperthermia and diaphoresis [2]. Neuromuscular symptoms are also usually observed, ranging from akathisia to muscle rigidity, hyperreflexia and myoclonus [2]. Serotonin syndrome can be a life-threatening condition if not promptly recognized and treated as it can progress to seizures or shock [1].
Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med 2005;352:1112-20.
Treatment
The most important initial step to the treatment of serotonin syndrome is to remove exposure to the offending agent. Use of activated charcoal to assist with gastrointestinal decontamination can be considered in the setting of a recent ingestion [1]. Following this, the cornerstone of therapy of serotonin syndrome is symptom based supportive care, with benzodiazepines being one of the most important components of treatment [1, 2]:
- Obtain an ECG to evaluate the QTc and QRS durations.
- Begin IV fluid rehydration and resuscitation.
- If the patient is hyperthermic, especially greater than 40 ºC, begin active external cooling.
- Antipyretics are not useful for the treatment of hyperthermia secondary to serotonin syndrome. This is because the hyperthermia is due to increased muscle activity and not secondary to change in the hypothalamic temperature set point.
- Benzodiazepines improve numerous symptoms of serotonin syndrome, especially agitation and are one of the most important elements of treatment.
- Use easily titrated medications like nitroprusside or esmolol for severe hypertension.
- If needed, vasopressors should be used for refractory hypotension.
- A recent study has suggested the potential utility of dexmedetomidine to treat serotonin syndrome (5).
- Cyproheptadine, a serotonin antagonist, has long been proposed anecdotally for the treatment of serotonin syndrome. However, multiple studies suggest that it does not change outcomes and may only help temporarily control symptoms (6).
Unless the clinical features are very mild, these patients will likely need monitoring in an ICU setting for at least 24 hours after being initially managed in the Emergency Department (ED). As always, especially in the case of an intentional overdose, contacting the local Poison Control Center is a valuable resource in helping to treat these patients.
What about Neuroleptic Malignant Syndrome?
One of the disorders that can present similarly to serotonin syndrome is neuroleptic malignant syndrome (NMS). Pathophysiologically, NMS is related to an inherited genetic mutation in skeletal muscle and is provoked in the presence of certain neuroleptics [7]. Neuroleptic malignant syndrome often occurs after exposure to drugs that affect the central dopaminergic system, such as haloperidol. There are inherent differences between NMS and serotonin syndrome. Serotonin syndrome tends to present within 24 hours of exposure to the offending agent, whereas NMS will often be more delayed, presenting after 7 days of starting the neuroleptic [4]. Patients with serotonin syndrome will often be agitated and delirious while NMS patients will often have dysphagia, incontinence and increased secretions [4]. NMS will often have extrapyramidal side effects, muscle rigidity and rhabdomyolysis compared to the mydriasis, clonus and hyperreflexia of serotonin syndrome [4].
Return to the Case
As noted above, the patient in this case exhibited all the hallmark signs of serotonin syndrome, including agitation, restlessness, diaphoresis, mydriasis, hyperreflexia and clonus. In the emergency department, this patient was treated with multiple rounds of lorazepam and boluses of saline which helped to control his symptoms. This patient was admitted to the medical ICU and monitored for an additional 24 hours. After his symptoms and vital signs normalized, he was transferred to the floor and received the psychiatric evaluation and treatment that he needed. His hospital course was uncomplicated and he was transferred to an inpatient psychiatric treatment facility for further care.
Take Home Points
- Serotonin syndrome can be a life-threatening condition. Keep a high suspicion for this in patients on serotonergic agents.
- The hallmark signs and symptoms of serotonin syndrome are anxiety and restlessness, diaphoresis, mydriasis, clonus, hyperrflexia, tachycardia and hypertension.
- Treatment is mainly supportive care and includes intravenous fluids, benzodiazepines titrated to symptom control and blood pressure control as needed.
- Serotonin syndrome is often confused with NMS. Remember, NMS is in the setting of exposure to neuroleptics and tends to present later on with symptoms of extrapyramidal side effects, muscle rigidity and increased secretions.
- Local poison control centers are a great resource in helping manage the treatment of patients with serotonin syndrome and most will require at least 24 hours in an ICU setting after ED diagnosis and management.
Expert Commentary
Dear Dr. Stelter,
Thank you again for yet another exciting tox blog post! This case has a tremendous amount to dissect, but I want to focus on a few things. For this case, let’s talk about the tox history, the tox physical exam, and the “supportive care” recommendation. Really, this is the definition of bread and butter medical toxicology.
The patient history described above is incredibly common, and that makes sense! Patients with a known history of psychiatric disease have higher risk of having psychiatric emergencies and have access to psychiatric medications. So whether by misunderstanding the purpose of their medication or by intentionally trying to harm oneself, it is common that we as medical toxicologists have to consider the effect a patient’s psychiatric medications have had (or will have) on their current clinical state. So what I want to know out of this patient is what all he could have been exposed to and when. Here are examples of questions I ask:
- “When did you take this medication?” (Looking at the clock) “Ok. It’s 3pm. You took it 2 hours ago, so at 1pm you sat down and swallowed 30 pills?” In essence, I want the patient to describe exactly how he was exposed – this can also clue me into his intent.
- “Did you take anything else?” (Answer is commonly “no” – always dig deeper) “Are you on any other medication? When is the last time you took that medication? When you started to feel sick before you came into the emergency department, did you take anything else to help with your symptoms?”
- “This may sound silly to you, but I also care about anything else you are taking. Are you on any vitamins or diet pills or herbals or cleanses? Is there anything you have purchased and taken in the last week which was not food or a prescription medication we have already talked about?”
- “Do you do any drugs?” I purposely leave the word “illicit” out. I don’t really care about legality. I just want to make them better. “I have seen many patients use all sorts of things that make them feel as sick as you feel now. Is there anything you can think of that you’ve taken which could make you this sick?”
This physical examination gave you your diagnosis! Usually we are clued into these diagnoses based on hyperthermia, which this patient frustratingly did not have. But that “hot tox” differential is something all emergency physicians should be aware of and facile in differentiating between. It includes serotonin syndrome, NMS, sympathomimetic toxicity (including cocaine, amphetamines, MDMA, cathinone derivatives, etc.), anticholinergic toxicity, and severe salicylate toxicity just to name a few. Although this patient did not have hyperthermia, he did have almost every other finding associated with serotonin syndrome with the most specifically-associated being clonus. Once I see clonus, I take a step back and see if serotonin syndrome fits into the clinical picture.
Finally, I greatly appreciate your description of symptom-focused supportive care in this clinical scenario. As you have displayed, “supportive care” is not super simple. It is nuanced and multifaceted and very frequently life-saving. Before fellowship I was frustrated that there wasn’t necessarily a specific antidote or procedure we could do to reverse toxicity in all poisoned patients. With time, however, I have gained great respect for “supportive care” – regretting when it is not performed adequately and truly appreciating when it is life-saving.
So in this single case you have touched on the key major aspects of the specialty of medical toxicology. I hope some of my points were helpful to you in further appreciating the stellar medical care you provided this sample patient.
Sincerely,
Patrick Lank, MD
Associate Program Director Northwestern Emergency Medicine
Assistant Professor of Emergency Medicine
Posts You May Also Enjoy
How to cite this post
[Peer-Reviewed, Web Publication] Stelter J, Byrne E (2017, Nov 13). Serotonin Syndrome [NUEM Blog. Expert Commentary By Lank P]. Retrieved from http://www.nuemblog.com/blog/serotonin-syndrome
References
- Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med 2005;352:1112-20.
- LoVecchio F, Mattison E. Atypical and serotonergic antidepressants. In Tintinalli’s emergency medicine: A comprehensive study guide. J Tintinalli (Ed.) (pp. 1219-24). 2016. New York, NY: McGraw-Hill.
- Pedavally S, Fugate JE, Rabinstein AA. Serotonin syndrome in the intensive care unit: Clinical presentations and precipitating medications. Neurocrit Care 2014;21:108-13.
- Birmes P, Coppin D, Schmitt L, Lauque D. Serotonin syndrome: A brief review. Can Med Assc J 2003;168:1439-42.
- Rushton WF, Charlton NP. Dexmedetomidine in the treatment of serotonin syndrome. Ann Pharmacotherapy 2014;14:1651-4.
- McDaniel WW. Serotonin syndrome: Early management with cyproheptadine. Ann Pharmacother 2001;35:870-3.
- Keck PE, Caroff SN, McElroy SL. Neuroleptic malignant syndrome and malignant hyperthermia: End of a controversy? J Neuropsychiatry Clin NeuroSci 1995;7:135-44.
Managing Alcohol Withdrawal In The ED
Over 16.3 million adults have an alcohol use disorder as defined by the NIH. Though the emergency department (ED) sees its fair share of intoxicated patients, many patients also present to the ED with withdrawal symptoms. This week we cover a few tips on management.
Beta Blocker Toxicity
Beta-blockers are the fifth most commonly prescribed medication in US and are frequently involved in potentially fatal single or multi-drug ingestions. Cardiovascular drugs as a whole are the 2nd most common culprit of adult death from all overdoses.
Button Battery Ingestion
According to Poison Control, about 3,300 button battery ingestions occur each year in the United States, with about two thirds of those cases occurring in patients younger than 6 years old.



























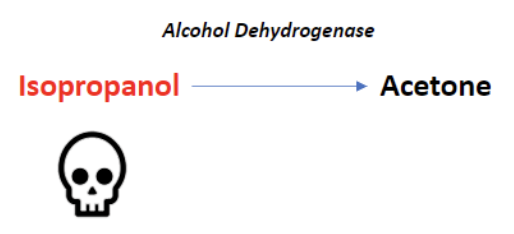












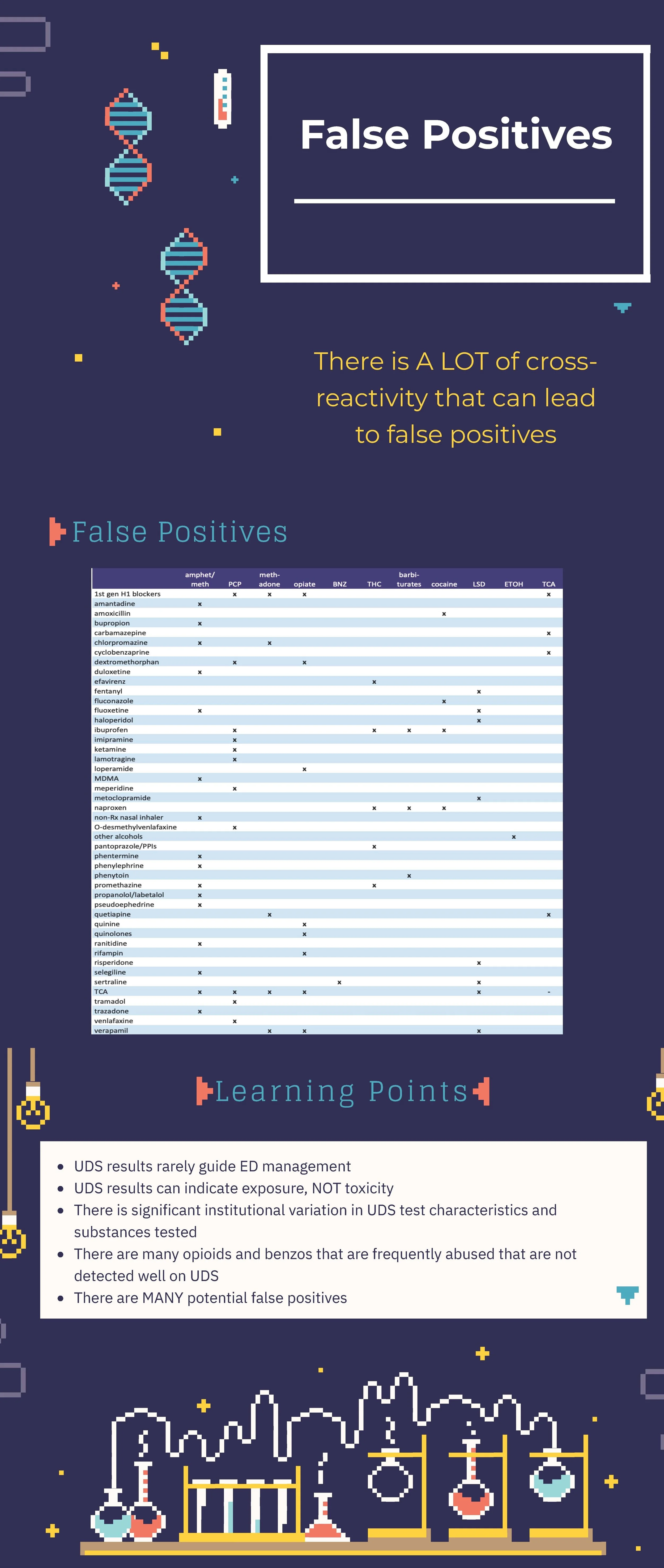





![Figure 1: Composition of Various Brands of Lipid Emulsions[1]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1594923133383-9YSM79RGG2VRCXN045S4/Intralipid+image+1.png)
![Figure 2: Maximum Doses and Durations of Various Local Anesthetics[9]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1595368949352-3SYZ5X29MWCMBS3CV8BT/Screen+Shot+2020-07-21+at+5.02.19+PM.png)