An overview of the diagnosis, management, and disposition of patients who present to the emergency department with acute diverticulitis.
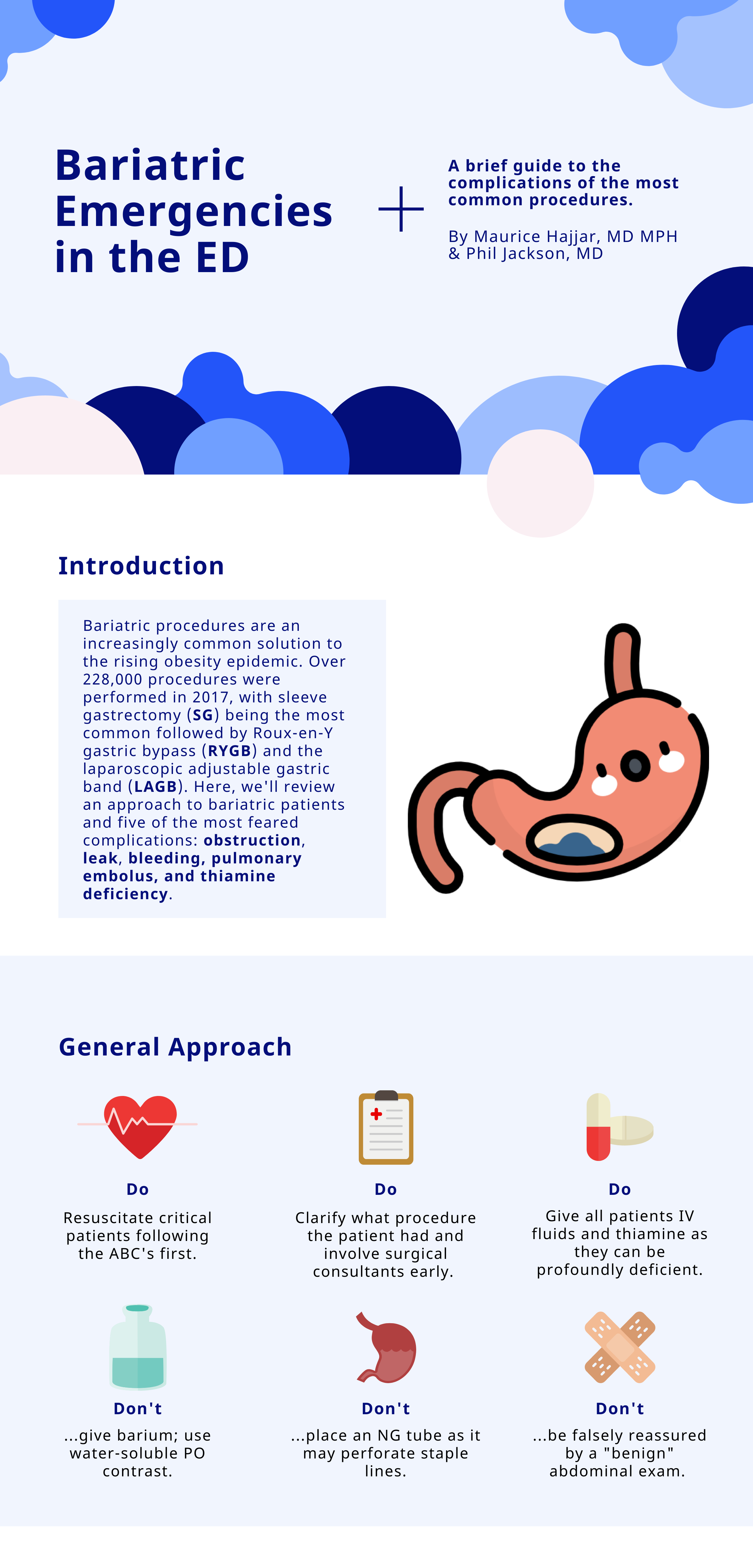
Bariatric Emergencies
Written by: Maurice Hajjar, MD, MPH (NUEM ‘22) Edited by: Philip Jackson(NUEM ‘20) Expert Commentary by: Gabby Ahlzadeh, MD
Expert Commentary
Thanks for this great post. Being familiar with the anatomy of these various procedures is essential to understanding the complications and why you cannot be reassured by a benign abdominal examination.
With laparoscopic band procedures, complications are more common early on and are more related to band erosion or migration. Migration of the lap-band is best evaluated with an upper GI series with Gastrografin. Another important question to ask patients is if their lap band has been inflated recently. This is typically done in a progressive manner, where normal saline is instilled within the subcutaneous port. This can also be a source of obstruction, and if emergent, the band can be deflated by aspirating fluid from the port. This should be done under the guidance of a surgeon, if possible.
Asking about dietary indiscretions can also sometimes clue you in to why a person is having abdominal pain or nausea. Especially immediately post-op, these patients have specific dietary guidelines and are often limited to liquids or pureed foods. Dumping syndrome can also occur in this patient population, more common with gastric bypass surgery, due to rapid gastric emptying. Typically, these patients have bloating, sweating, facial flushing, diarrhea, nausea, early satiety about 30 to 60 minutes after a meal. Dumping syndrome is typically diagnosed clinically, though laboratory testing should be performed to rule out electrolyte derangements. Dumping syndrome is typically treated with dietary modifications and should be discussed with the patient’s surgical team.
As these procedures become more common and advanced, some are also performed endoscopically, which comes with its own set of complications. Another newer procedure is the intragastric balloon, which is a saline filled silicone balloon that is inflated in the stomach. In the first few days after placement, patients may experience abdominal pain, nausea, and vomiting. Other complications include balloon rupture, bowel obstruction, gastric outlet obstruction, gastric ulcer, pancreatitis, nonalcoholic steatohepatitis and cholecystitis.
Overall, management of these patients in the emergency department should be done in consultation with the surgeon.
Gabrielle Ahlzadeh, MD
Clinical Assistant Professor of Emergency Medicine
University of Southern California
How To Cite This Post:
[Peer-Reviewed, Web Publication] Hajjar, M. Jackson, P. (2021, Apr 26). Bariatric Emergencies. [NUEM Blog. Expert Commentary by Ahlzadeh, G]. Retrieved from http://www.nuemblog.com/blog/bariatric-emergencies
Other Posts You May Enjoy
A Practical Approach to Abdominal Imaging
Written by: Zach Schmitz MD (PGY-3) Edited by: David Kaltman, MD (PGY-4) Expert commentary by: Samir Abboud, MD
I often find myself in a gray zone when it comes to imaging abdominal pain. Any third year medical student worth their salt can tell you to get the RUQ ultrasound for the fat, fertile, forty year-old female with RUQ abdominal pain, fever, positive Murphy’s sign, and leukocytosis. However, my patients don’t usually fit the textbook, and I’m often thinking about what I might miss or see with test X vs test Y. Below, I’ll touch on a few common dilemmas where the optimal choice of imaging modality isn’t immediately clear by focusing on what you actually gain or lose by ordering one imaging test over another.
Scenario 1: Stone or Appendicitis?
Case: 62 year old female with HTN and HLD presents with RLQ pain. The pain woke her this morning and has been intermittent all day, occurring exclusively when she urinates. It is sharp, non-radiating, and increasing in intensity. She never had a pain like this and can now barely sit still. She has thrown up a few times over the past few hours. Vitals are stable and she is afebrile. She appears uncomfortable with RLQ tenderness but no rebound or guarding. Labs show slight leukocytosis, and urine has no blood.
If I suspect stone over appendicitis, will a CT without contrast miss appendicitis?
CT, MR, and US are well studied in their ability to detect and accurately diagnose appendicitis.[1]
CT with IV contrast is 96-100% sensitive and 91-100% specific. Per the American College of Radiology’s (ACR) appropriateness system, this is the most appropriate initial test for suspected appendicitis in adults.[2]
MR is 96% sensitive and 96% specific.[3]
Ultrasound has a wide range of data, with sensitivity ranging from 21-95.7% and specificity of 71-97%.[2]
CT without oral or IV contrast is nearly as useful for diagnosing appendicitis
A meta-analysis by Xiong et al included seven original studies investigating a total of 845 patients.[4]
Pooled sensitivity - 0.90 (95% CI: 0.86-0.92)
Pooled specificity - 0.94 (95% CI: 0.92-0.97)
Pooled positive likelihood ratio - 12.90 (95% CI: 4.80-34.67)
Pooled negative likelihood ratio - 0.09 (95% CI: 0.04-0.20)
Will a contrast enhanced CT for appendicitis ruin my chance to catch a kidney stone?
Non-contrast CT is the emergency standard in diagnosing nephrolithiasis with good reason - it is 97% sensitive and 95% specific.[5]
Will contrast ruin the ability to detect a stone?
This makes theoretical sense as stones and contrast are both hyper-intense on CT.
Sensitivity is decreased for small stones with contrast enhanced studies.
However, for stones > 3mm, sensitivity remains 95%.[5]
Only about 5% of stones that small ultimately require intervention.
Takeaways: You sacrifice a bit with a non-contrast study looking for appendicitis and a contrast enhanced study looking for stone, but both still work well. The American Urology Association recommends consultation for stones > 10mm.[6] Urology would also need to be involved with signs of sepsis, abscess, deterioration in renal function, intractable symptoms, or a transplant/solitary kidney. It seems I am very likely to see a stone requiring something other than watchful waiting on a CT with contrast. It is worse to miss an appendicitis than a 2mm stone, so contrast might make more sense if it’s close.
Scenario 2: RUQ Ultrasound after Negative CT San
Case: 84 year old male with a history of prostate cancer and hypertension presents from a nursing home with 4 days of diffuse abdominal pain. He has had no vomiting or bowel movements over this time. No urinary symptoms. He is hemodynamically stable, and his abdomen is diffusely tender (maybe worse in the RUQ) and distended but overall not terribly impressive. You order a CT for possible obstruction and it just shows a large stool burden. The gallbladder was visualized and looked normal.
If a CT is negative, should I get a RUQ US to look for cholecystitis?
RUQ Ultrasound
Per ACR, this is the most appropriate initial study for RUQ pain and suspected biliary disease.[7]
A 2012 meta analysis showed a sensitivity of 81% (95% CI 75-87%) and specificity of 88% for acute cholecystitis.[8]
It has the advantage of being dynamic, with a sonographic Murphy sign independently showing an 86% sensitivity and 35% specificity, positive predictive value of 43%, and negative predictive value of 82%.[9]
Computed Tomography (CT)
The same 2012 meta analysis only had one study with CT, but noted a sensitivity of 94% with fairly broad confidence intervals (95% CI 73-99) and a specificity of only 59%.[8]
ACR notes CT’s NPV for acute cholecystitis approaches 90%.[7]
A 2015 study looked at 101 patients who went to the OR and got both a CT and US. For acute cholecystitis, the sensitivities for CT and US were 92% and 79% respectively. For cholilithiasis, sensitivities for CT and US were 60% and 89% respectively.[10,11]
ACR states it is “usually appropriate” to proceed with CT for RUQ pain and suspected biliary disease with a negative or equivocal ultrasound.[7]
Although it lacks a sonographic murphy’s sign equivalent, its advantage is to help in operative planning and seeing complications, such as perforation or gangrene.
MRI has a sensitivity of 85% and a specificity of 81%. It is also considered “usually appropriate” by ACR if ultrasound is negative or equivocal[7]
Cholescintigraphy is the best imaging, showing 97% sensitivity and 90% specificity for acute cholecystitis. It is also the most appropriate study if you suspect acalculous cholecystitis.[7]
Takeaways: There are a few interesting points from this set of data. First, CT seems to have at least as good of ability to pick up cholecystitis compared to ultrasound. However, it is much worse in detecting gallstones themselves, which may be very relevant to a patient with abdominal pain. Second, the sensitivity of both RUQUS or CT isn’t really that great and we are probably missing a few episodes of cholecystitis. If there is a very high index of suspicion but negative imaging, it may be worthwhile to pursue additional workup. Overall, if the CT shows a normal gallbladder, and you are not worried about intractable biliary colic, the ultrasound probably won’t add much.
Scenario 3: Female Pelvic Pain
Case: 33 year old female with a history of chlamydia infection presenting with right sided abdominal pain. The pain has gradually been getting worse for 1 day. She has had a few episodes of vomiting. There is some white vaginal discharge she always has. On exam, she is tachycardic, normotensive, and febrile to 101.5. She has RLQ tenderness with voluntary guarding. On pelvic exam, there is some white vaginal discharge, CMT, R adnexal tenderness that seems less intense than her RLQ tenderness, and no masses noted.
If this patient had a normal appendix and ovaries after a contrast enhanced CT for appendicitis, how useful is an additional transvaginal ultrasound to rule out gynecologic pathologies?
For ovarian torsion:
A retrospective study of 834 patients showed the NPV of a contrast enhanced CT of the pelvis for ovarian torsion is 100%.[12]
A prospective study of 199 patients showed doppler ultrasound has a sensitivity and specificity for torsion of 100 and 97%.[13]
For Tubo-Ovarian Abscess (TOA):
CT is thought to be between 78 and 100% sensitive.[14]
2011 literature review gives a broad range of sensitivity and specificity for US in TOA with a sensitivity of 56-93% and specificity from 89-98%.[15]
Takeaways: ACR appropriates rates ultrasound as the most appropriate test for female pelvic pain.[14] However, it also rates CT with contrast as more appropriate for suspected appendicitis.[2] This patient raises concerns for both, and a CT was done first. CT is good for finding intra abdominal and pelvic abscess. It is more difficult to assess how useful ultrasound is for TOA, as many studies in the literature review were either before year 2000 or used a transabdominal approach. Overall, if someone has a CT scan for appendicitis that shows normal ovaries, the transvaginal ultrasound seems to add little for either torsion or TOA.
One potential dangerous conclusion from this set of data is that we should just CT everyone up front. While CT shows good sensitivities for many of the pathologies in question, simply ordering a CT first ignores the many good reasons - such as cost, radiation dose, speed, improved specificity and comparable sensitivity, resource utilization, sonographic murphy sign - RUQUS and pelvic ultrasound are the most appropriate initial tests for suspect biliary and pelvic pathology. That said, it a patient has an entirely normal CT that was already performed for other indicated reasons, the use of additional imaging may be unnecessary and should be considered carefully. Overall, the question of exactly what imaging test to order when ruling out common, emergent, abdominal pathologies is often a difficult one with shades of gray. By having a better understanding of exactly what type of information we are getting and missing from each test we order, emergency physicians can more quickly, safely, and accurately diagnose and treat our patients.
Expert Commentary
This is a thoughtful, well-reasoned approach to optimizing the imaging strategy in challenging, atypical clinical scenarios. To add a few nuances to some of the points raised:
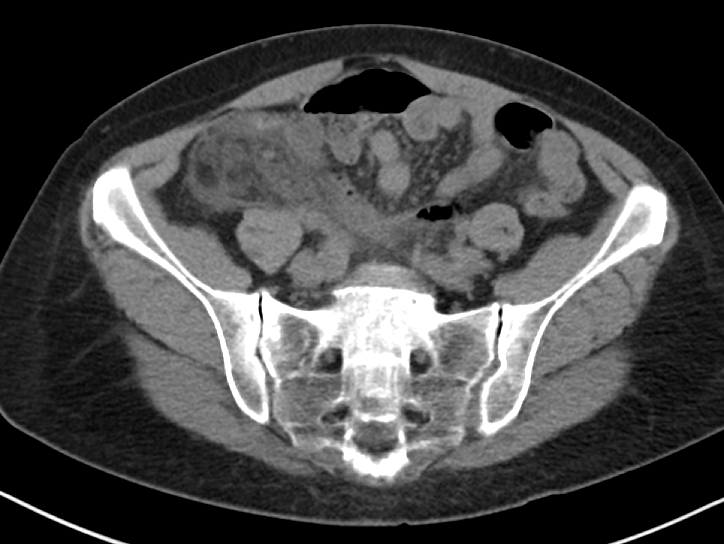
When considering a contrast-enhanced versus non-contrast CT (both IV and PO) in the clinically ambiguous scenario, it is important to consider your patient’s body habitus. Figure 1 includes representative images from a non-contrast enhanced CT of a patient with a BMI above 25. You can clearly see the inflammatory stranding in the right lower quadrant mesenteric fat (Figure 1a) and portions of an appendicolith (Figure 1 b), in this patient who ultimately proved to have appendicitis. The natural contrast provided by the patient’s mesenteric fat in this scenario helps us work around the absence of IV contrast.
Figure 1a
Figure 1b
Figure 2 includes representative images from a contrast enhanced CT of a very thin patient, with a relative paucity of intra-abdominal fat. In this patient, the relative absence of natural contrast would greatly reduce our chances to diagnose appendicitis (or even identify the appendix) in the absence of IV contrast. PO contrast is additionally likely to be most helpful in very thin patients [Alabousi 2015].
Figure 2
The author asks (and answers) a very insightful question with regards to identifying kidney stones on contrast enhanced CT. A few points to add:
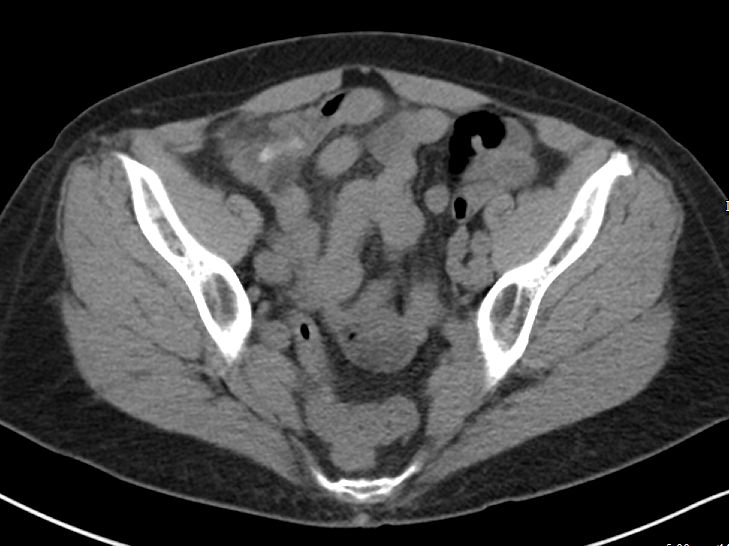
Assuming the contrast enhanced study is obtained prior to the excretory phase of imaging (and most routine studies are) ureteral stones should still be largely visible - the stones that will generally be more difficult to identify will be the non-obstructing stones still within the collecting system. Additionally, while there is indeed a small sacrifice in sensitivity for small stones with contrast enhanced studies, the identification of secondary complications is much improved.
Consider Figure 3, which demonstrates a 2 mm stone in the proximal left ureter identified on a contrast enhanced study. Notice the slightly delayed nephrogram on the left relative to the right, which could indicate a component of obstructive uropathy. Similarly, identification of such complications as pyonephrosis, pyelonephritis, and perinephric abscess is much improved with contrast enhanced images. For this reason, I would suggest that in the clinically ambiguous scenario, erring on the side of the contrast enhanced study would be wise.
Figure 3
It is important to note that the CT scanner installed in our emergency department is a dual-energy machine. Many of our other departmental scanners are dual-energy as well. With these scanners, we are able to apply algorithms to deconstruct the elemental composition of stones and provide more information than simply size and location - i.e. uric acid or non-uric acid stone - if requested. We can additionally generate virtual non-contrast images from the contrast-enhanced images, without exposing our patients to additional radiation. While it is tempting to think that we could recapture some of the sensitivity for renal stones using these virtual non-contrast images, this has unfortunately not been borne out in the literature at this time [Vrtiska 2010], though remains an area of continued investigation as imaging technology is further improved.
The advantages of dual-energy imaging are not only limited to the kidneys. With regards to the evaluation of biliary colic, virtual monochromatic images can be generated with resulting increased conspicuity of gallstones, even those that appear isodense to bile on the conventional images [Ratanaprasatporn 2018].
In general, if you find yourself with a high degree of suspicion for any disease process and discordant imaging findings, I would encourage you to call your radiologist. The additional clinical information exchanged during such a call may direct what additional data sets should be generated and what additional imaging studies may be of most benefit. Last, but certainly not least, that “second look” armed with additional clinical information can pick up on subtle findings that are, in isolation, entirely non-specific, but in a certain clinical scenario could clinch the diagnosis you are seeking.
References:
Alabousi A et al. Is Oral Contrast Necessary for Multidetector Computed Tomography Imaging of Patients With Acute Abdominal Pain? Canadian Association of Radiologists Journal. 2015;66(4): 318 - 322
Ratanaprasatporn L et al. Multimodality Imaging, including Dual-Energy CT, in the Evaluation of Gallbladder Disease. Radiographics 2018;38(1): 75-89
Vrtiska TJ et al. Genitourinary Applications of Dual-Energy CT. American Journal of Roentgenology. 2010;194: 1434-1442.
Samir Abboud, MD
Assistant Professor of Radiology
Northwestern University
How To Cite This Post:
[Peer-Reviewed, Web Publication] Schmitz, Z. Kaltman, D. (2020, Feb 10). An Approach to Abdominal Imaging. [NUEM Blog. Expert Commentary by Abboud, S]. Retrieved from http://www.nuemblog.com/blog/abdominal-imaging.
Other Posts You Might Enjoy…
References
Dahabreh IJ, Adam GP, Halladay CW, Steele DW, Daiello LA, Weiland LS, Zgodic A, Smith BT, Herliczek TW, Shah N, Trikalinos TA. Diagnosis of Right Lower Quadrant Pain and Suspected Acute Appendicitis. Comparative Effectiveness Review No. 157. (Prepared by the Brown Evidence-based Practice Center under Contract No. 290-2012-00012-I.) AHRQ Publication No. 15(16)-EHC025-EF. Rockville, MD: Agency for Healthcare Research and Quality; December 2015. www.effectivehealthcare.ahrq.gov/reports/final.cfm.
American College of Radiology. ACR Appropriateness Criteria®: RLQ pain. Available at https://acsearch.acr.org/docs/69357/Narrative/ Accessed 5/10/19.
Duke E, Kalb B, Arif-Tiwari H, et al. A Systematic Review and Meta-Analysis of Diagnostic Performance of MRI for Evaluation of Acute Appendicitis. AJR Am J Roentgenol 2016;206:508-17.
Xiong B, Zhong B, Li Z, Zhou F, Hu R, Feng Z, Xu S, Chen F. Diagnostic Accuracy of Noncontrast CT in Detecting Acute Appendicitis: A Meta-analysis of Prospective Studies. Am Surg. 2015 Jun;81(6):626-9.
Curhan G, Aronson M, Preminger G. Diagnosis and acute management of suspected nephrolithiasis in adults. UpToDate.com. April 30 2019.
Assimos D, Krambek A, Miller N et al. Surgical Management of Stones: AUA/Endourology Society Guideline (2016). https://www.auanet.org/guidelines/kidney-stones-surgical-management-guideline. Accessed 5/10/19.
American College of Radiology. ACR Appropriateness Criteria®: RUQ pain. Available at https://acsearch.acr.org/docs/69474/Narrative/ .
Kiewiet J.J., Leeuwenburgh M.M., Bipat S., et al: A systematic review and meta-analysis of diagnostic performance of imaging in acute cholecystitis. Radiology 2012; 264: pp. 708-720.
Bree, Robert L. Further observations on the usefulness of the sonographic Murphy sign in the evaluation of suspected acute cholecystitis. Journal of Clinical Ultrasound. March/April 1995.
Wertz JR1,2, Lopez JM3, Olson D4, Thompson WM1,2. Comparing the Diagnostic Accuracy of Ultrasound and CT in Evaluating Acute Cholecystitis. AJR Am J Roentgenol. 2018 Aug;211(2):W92-W97. doi: 10.2214/AJR.17.18884. Epub 2018 Apr 27.
Fagenholz, P et al. Computed Tomography Is More Sensitive than Ultrasound for the Diagnosis of Acute Cholecystitis. Surg Infect (Larchmt). 2015 Oct;16(5):509-12. doi: 10.1089/sur.2015.102. Epub 2015 Sep 16.
Lam A1, Nayyar M2, Helmy M2, Houshyar R2, Marfori W2, Lall C2.Assessing the clinical utility of color Doppler ultrasound for ovarian torsion in the setting of a negative contrast-enhanced CT scan of the abdomen and pelvis. Abdom Imaging. 2015 Oct;40(8):3206-13. Doi: 10.1007/s00261-015-0535-4.
Laufer, M. Ovarian and fallopian tube torsion. UpToDate. April 30 2019. https://www.uptodate.com/contents/ovarian-and-fallopian-tube-torsion?search=ovarian%20torsion&source=search_result&selectedTitle=1~70&usage_type=default&display_rank=1 .
Beigi, R. Epidemiology, clinical manifestations, and diagnosis of tubo-ovarian abscess. UpToDate. April 30 2019. https://www.uptodate.com/contents/epidemiology-clinical-manifestations-and-diagnosis-of-tubo-ovarian-abscess?search=tuboovarian%20abscess&source=search_result&selectedTitle=2~24&usage_type=default&display_rank=2 .
Lee DC1, Swaminathan AK. Sensitivity of ultrasound for the diagnosis of tubo-ovarian abscess: a case report and literature review. J Emerg Med. 2011 Feb;40(2):170-5. doi: 10.1016/j.jemermed.2010.02.033. Epub 2010 May 13.
American College of Radiology. ACR Appropriateness Criteria®: Female Pelvic Pain. Available at https://acsearch.acr.org/docs/69503/Narrative/ .
Spontaneous Bacterial Peritonitis
Written by: Kevin Dyer, MD (NUEM PGY-4) Edited by: Paul Trinquero, MD (NUEM Alum ‘19) Expert commentary by: Steven K. Herrine, MD
Background
Spontaneous bacterial peritonitis (SBP) is an infection of ascitic fluid without an evident intra-abdominal or surgically-treatable source [1]. The pathophysiology is poorly understood, but likely relates to bacterial translocation across an edematous bowel wall (due to portal hypertension) into the immunologically impaired ascitic fluid. SBP is common in patients with severe hepatic dysfunction (present in 20% of hospitalized patients with ascites) and carries a mortality rate of 10% [2,3]. The high incidence, grave mortality, and potential benefit of early treatment make SBP a can’t-miss diagnosis for the emergency physician.
Clinical Features
The classic triad of SBP is fever, abdominal pain, and increasing ascites [4]. However, like most other “classic triads” in medicine, patients rarely present with all three and instead are much more likely to have only 1 or 2 of these features. The most common signs and symptoms are fever (69%), abdominal pain (59%), altered mental status (54%), and abdominal tenderness (49%). Other less common signs and symptoms include diarrhea (32%), paralytic ileus (30%), hypotension (21%), and hypothermia (17%) [5]. In one study, 13% of patients with SBP were asymptomatic [6].
Importance of Paracentesis
As mentioned above, the clinical signs and symptoms of SBP are variable and sometimes subtle, making it a difficult clinical diagnosis. Instead, diagnosis hinges on the results of an ascitic fluid sample obtained via a diagnostic paracentesis. Ascitic fluid findings indicative of SBP include an elevated absolute polymorphonuclear leukocyte (PMN) count ≥250 cells/mm3, a positive ascitic fluid bacterial culture, and exclusion of secondary causes of bacterial peritonitis (e.g. perforated viscous, intra-abdominal abscess). For the ED physician, a PMN level ≥250 cells/mm3 necessitates initiation of antibiotics as soon as possible, if they were not already started empirically [7].
Not only do patients at risk for SBP need a diagnostic paracentesis, but because treatment is time sensitive, it should be performed as quickly as is feasible. A study by Kim et al showed a 3.3 percent increase in in-hospital mortality for each hour paracentesis was delayed and revealed that delayed paracentesis for patients ultimately diagnosed with SBP led to a 2.7-fold increased risk of death [8-9].
Treatment
Empiric therapy should be initiated in patients with ascites who have a temperature >100°F, abdominal pain and/or tenderness, altered mental status, or ascitic PMNs ≥250 cells/mm3. First line treatment is a third-generation cephalosporin such as Ceftriaxone 2g per day or Cefotaxime 2g q8hrs [10]. For those with a severe penicillin allergy, fluoroquinolones are a viable alternative provided the patient has not been on a prophylactic fluoroquinolone. Common choices are levofloxacin 750mg daily or ofloxacin 400mg BID [10-11].
An additional therapeutic consideration is albumin infusion. Current guidelines from the American Association for the Study of Liver Diseases (AASLD) recommend the administration of albumin at a dose of 1.5g/kg for patients with either a BUN >30mg/dL, a serum creatinine >1.30mg/dL, or a total bilirubin >4mg/dL. A meta-analysis of 4 studies demonstrated a significant reduction in renal impairment (8% vs 31%) and a decrease in mortality (16% vs 35%) in patients who received 1.5g/kg of albumin within 6 hours of diagnosis of SBP [12-13].
Take Home Points
Spontaneous bacterial peritonitis is an infection of ascitic fluid without an evident intra-abdominal or surgically-treatable source.
Diagnosis is nearly impossible to make clinically due to variable, and sometimes subtle, presenting symptoms and therefore hinges on obtaining a fluid sample via a diagnostic paracentesis.
Early diagnostic paracentesis in the ED may improve mortality for hospitalized patients with ascites.
Polymorphonuclear leukocyte (PMN) count ≥250 cells/mm3 raises suspicion for SBP and should prompt initiation of empiric antibiotics - first line is 2g of Ceftriaxone and second line is a fluoroquinolone.
In addition, AASLD guidelines recommend 1.5g/kg of albumin for patients with SBP who have BUN >30mg/dL, creatinine >1.3mg/dL, or bilirubin >4mg/dL as this may decrease mortality and reduce the incidence of renal impairment.
Expert Commentary
The blog authors make many excellent points about the recognition and management of SBP in the emergency department. From the standpoint of a hepatologists, several concepts and developments regarding this dangerous diagnosis are worth emphasizing.
Clinical context is vital in making management decisions: a patient with advanced chronic liver disease, manifested by high MELD score, is more likely to have SBP than an individual with acute or less advanced disease.
Prompt paracentesis is the key to proper management. It is considered safe to perform diagnostic paracentesis in all patients but those with evident hyperfibrinolysis or those in DIC. Some have advocated for midline paracentesis to avoid vascular structures, but LLQ has been demonstrated to be safer. (Sakai 2005)
Only a small amounts of fluid is required to make the diagnosis of PMN > 250 cells/mL. Even a drop in a hemocytometer can be sufficient
Patients on prophylactic antibiotics due to previous SBP or low-protein ascites should raise concern for resistant organisms. It is important to sample the ascites in these individuals before commencing antibiotic therapy.
The use of dipstick (leukocyte esterase) diagnosis is supported by some literature but use has been limited mostly to developing nations (Mendler 2010)
There is a growing body of literature that supports initial intravenous therapy followed by transition to oral antibiotics in clinical scenarios meeting certain conditions. (Angeli 2006)
References:
Sakai H, Sheer TA, Mendler MH, Runyon BA. Choosing the location for non-image guided abdominal paracentesis. Liver International 2005;25:984-986.
Mendler MH, Agarwal A, Trimzi M, Magridal E, Tsushima M, Joo E, Santiago M, et al. A new highly sensitive point of care screen for spontaneous bacterial peritonitis using a leukocyte esterase method. J Hepatol 2010;53:477-483
Angeli P, Guarda S, Fasolato S, Miola E, Craighhero R, Del Piccolo F, Antona C, et al. Switch therapy with ciprofloxacin vs intravenous ceftazidime in the treatment of spontaneous bacterial peritonitis in patients with cirrhosis: similar efficacy at lower cost. Aliment Pharmacol Ther 2006;23:75-84
Steven K. Herrine, MD
Professor of Medicine, Division of Gastroenterology and Hepatology
Vice Dean for Academic Affairs
Sidney Kimmel Medical College at Thomas Jefferson University
How to Cite this Post
[Peer-Reviewed, Web Publication] Dyer K, Trinquero P. (2019, July 29). Spontaneous Bacterial Peritonitis. [NUEM Blog. Expert Commentary by Herrine S]. Retrieved from http://www.nuemblog.com/blog/sbp.
Other Posts You May Enjoy
References:
Such J, Runyon BA. Spontaneous bacterial peritonitis. Clin Infect Dis 1998; 27:669.
Nickson, Chris. “Spontaneous Bacterial Peritonitis.” Life in the Fast Lane Medical Blog, 17 Dec. 2012, https://www.lifeinthefastlane.com/ccc/spontaneous-bacterial-peritonitis
Guiney, Allan. “Should All Admitted Patients with Ascites Get a Paracentesis?” Core EM, 7 Dec. 2017, coreem.net/journal-reviews/paracentesis-for-all
“Episode 123.0 – Paracentesis Journal Update.” Core EM, 27 Nov. 2017, coreem.net/podcast/episode-123-0
McHutchison JG, Runyon BA. Spontaneous bacterial peritonitis. In Surawicz CM, Owen RL (Eds.), Gastrointestinal and Hepatic Infections, WB Saunders Company, Philadelphia 1995. P.455.
Runyon BA. Monomicrobial nonneutrocytic bacterascites: a variant of spontaneous bacterial peritonitis. Hepatology 1990; 12:710Runyon BA. Monomicrobial nonneutrocytic bacterascites: a variant of spontaneous bacterial peritonitis. Hepatology 1990; 12:710.
Runyon, B. (2018) Spontaneous bacterial peritonitis in adults: Clinical manifestations. In K.D. Lindor (Ed.), UpToDate. Retrieved April 2, 2018, from https://www.uptodate.com/contents/spontaneous-bacterial-peritonitis-in-adults-clinical-manifestations
Kim JJ, Tsukamoto MM, Mathur AK, et al. Delayed paracentesis is associated with increased in-hospital mortality in patients with spontaneous bacterial peritonitis. Am J Gastroenterol 2014; 109:1436.
Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006; 34:1589.
Runyon, B. (2018) Spontaneous bacterial peritonitis in adults: Treatment and prophylaxis. In K.D. Lindor (Ed.), UpToDate. Retrieved April 2, 2018, from https://www.uptodate.com/contents/spontaneous-bacterial-peritonitis-in-adults-treatment-and-prophylaxis
Navasa M, Follo A, Llovet JM, et al. Randomized, comparative study of oral ofloxacin versus intravenous cefotaxime in spontaneous bacterial peritonitis. Gastroenterology 1996; 111:1011.
Salerno F, Navickis RJ, Wilkes MM. Albumin infusion improves outcomes of patients with spontaneous bacterial peritonitis: a meta-analysis of randomized trials. Clin Gastroenterol Hepatol 2013; 11:123.
Pescatore, Rick. “Should You Give Albumin in Spontaneous Bacterial Peritonitis (SBP)?” R.E.B.E.L. EM - Emergency Medicine Blog, 6 July 2017, rebelem.com/should-you-give-albumin-in-spontaneous-bacterial-peritonitis-sbp
Inguinal Hernia Imaging and Reduction
Written by: Jesus Trevino MD, MBA (NUEM PGY-3) Edited by: Samia Farooqi, MD (NUEM 2016 Graduate) Expert Commentary by: Adriana Segura Olson, MD
Case
Mr. FJ is a 84 year old male with history of bilateral inguinal hernias status post repair with mesh 26 years ago who presents with constipation. Five days ago, he suffered from a profuse diarrheal illness that also affected his household members. Two days ago, he developed constipation with gradually progressive abdominal bloating, belching, and today bilious emesis. On exam, he is afebrile, hemodynamically stable with moderate abdominal distention and right inguinal swelling that is tender, non-mobile and without overlying skin changes.
A CT abdomen and pelvis revealed a small bowel obstruction secondary to a right-sided incarcerated inguinal hernia!
Types of abdominal hernias
Abdominal hernias can be classified based on etiology (i.e., congenital vs acquired) and location (Adams):
A retrospective review of 2510 hernia repairs from a single institution in Scotland (1985-2008) found the following distribution of abdominal hernias by location (Dabbas):
Inguinal (70.7%)
Umbilical (13.9%)
Epigastric (6.6%)
Incisional (4.7%)
Femoral (3.7%)
Spigelian/other (0.4)
Of inguinal hernias, the indirect form account for 65% of cases (Adams).
Clinical presentation and physical exam findings
Patient characteristics associated with an increased risk of abdominal hernia include (Fitzgibbons):
Male sex
Lower BMI
Family history of abdominal hernia
COPD
Smoking
Collagen vascular disease
Thoracic or abdominal aortic aneurysm
Open abdominal surgery
Peritoneal dialysis
On presentation, patients typically complain of abdominal or scrotal pain with or without superficial abdominal wall swelling that may worsen with maneuvers that increase intra-abdominal pressures (Adams). There may be a preceding event involving heavy lifting, coughing or other form of straining. (Adams). Symptoms such as nausea, emesis, constipation, and abdominal distention raise concern for small bowel obstruction.
On exam, inspection may reveal swelling in a ventral, umbilical, inguinal or femoral location. Often, the swelling can be reduced with gentle pressure - a reducible hernia; those that cannot be reduced at bedside are classified as incarcerated hernias. Tenderness to palpation and overlying skin changes, such as red, purple or blue coloration, increases suspicion for strangulated hernia.
Imaging
CT abdomen and pelvis is a good imaging modality to assess for abdominal hernia, especially when there is concern for acute incarceration or strangulation. CT findings include a “zone of transition” depicting a change in diameter of small bowel from dilated to a normal or decreased diameter such as the “pinch point” seen in the case image. Signs concerning for strangulation include engorged vessels within incarcerated hernia, fat stranding and thickened bowel wall (Strange).
A prospective study in Australia of patients with abdominal pain presenting to an ambulatory practice showed CT without contrast was 90% sensitive and 97% specific for the diagnosis of abdominal hernia, yielding a LR+ 26 and LR- 0.10 (Garvey). Assuming greater acuity of symptoms in the Emergency Department is associated with greater anatomical abnormalities more easily detected on CT, these test statistics can be generalized to the emergency setting.
Reduction
Bedside reduction is indicated when a hernia is incarcerated without evidence of strangulation. Signs suggestive of necrosis of hernia contents include peritonitis and erythema or necrosis of the overlying skin (Adams).
To prepare for reduction, place the patient supine in Trendelenburg (-20 degrees) with an ice pack overlying the area of swelling (Roberts). In addition to procedural sedation, immediate pain control soon after ED arrival facilitates abdominal wall muscle relaxation and increases likelihood of reduction success, sometimes even spontaneously when the patient is properly positioned.
Next, palpate the outline of the abdominal wall defect with the nondominant hand and place gentle inward pressure with the dominant hand at the base (i.e., the most proximal portion) of the hernia contents to slide the contents intra-abdominally. As the hernia contents slide through the abdominal wall defect, take care to avoid completely collapsing the lumen of the trapped small bowel at the “pinch point” as this will lead the remaining trapped bowel to distend (i.e., the ballooning effect), exceed the dimension of the wall defect and reduce the chances of a successful reduction at the bedside.
Disposition
Reducible abdominal wall hernias without signs of bowel ischemia can be discharged with appropriate outpatient general surgery follow-up for elective repair.
Hernias that remain incarcerated or have evidence of strangulation require general surgery consult for eventual OR reduction. Factors associated with difficult bedside reduction include duration of incarceration and small abdominal wall defect.
Case resolution
Bedside reduction failed; in retrospect, there was a low likelihood success based on the small “pinch point” (1.3 cm) relative to the bulk of the hernia contents. Mr. FJ was admitted and underwent open inguinal hernia repair with small bowel resection, primary anastomosis and discharged on post-operative day 14.
Expert Commentary
This is an excellent summary by Dr. Trevino of the Emergency Department management of hernias with respect to imaging and reduction.
As mentioned in this post, it is important to determine whether an incarcerated hernia is associated with strangulation, which indicates bowel necrosis. Hernias that are clearly strangulated should not be reduced at the bedside and surgical consultation is warranted; however, these are not always easily distinguishable on physical exam. While patients with incarcerated hernias without strangulation can present with systemic symptoms including nausea and vomiting, signs of strangulation include toxic appearance of the patient, significant systemic symptoms, or pain that persists after reduction of the hernia. A lactate is often sent in cases of an incarcerated hernia, but its sensitivity and specificity is limited and therefore, when there is a high index of suspicion, a normal lactate level should not be used to rule out strangulation (Derikx).
We often feel reassured with a normal lactate, but be wary of relying on this test and don’t let your surgical consultants rely too heavily on the lactate either!
It’s worth mentioning cases of internal hernias, which involve bowel protrusion through the peritoneum or mesentery into another compartment in the abdominal cavity. While rare, they are important to keep on the differential of undifferentiated abdominal pain as they can lead to small bowel obstruction or bowel necrosis. Internal hernias can be difficult to diagnose clinically because their presentation is often vague and intermittent, and they are not palpable on physical exam. CT scan of the abdomen and pelvis is the diagnostic modality of choice and this imaging may be normal in cases of reducible internal hernias. When diagnosed, prompt surgical consultation is indicated.
Adriana Segura Olson, MD
Assistant Professor, UT San Antonio Department of Emergency Medicine
Posts You May Also Enjoy
How to cite this post
[Peer-Reviewed, Web Publication] Trevino J, Farooqi S (2017, Aug 15). Inguinal Hernia Imaging and Reduction. [NUEM Blog. Expert Commentary By Olson AS]. Retrieved from http://www.nuemblog.com/blog/hernia-reduction
References
Adams, James. Emergency Medicine: Clinical Essentials. Philadelphia, PA: Elsevier/Saunders, 2013. Print.
Dabbas, N., K. Adams, K. Pearson, and G. Royle. "Frequency of Abdominal Wall Hernias: Is Classical Teaching out of Date?" JRSM Short Reports 2.1 (2011): 5. Print.
Garvey, J. F. W. "Computed Tomography Scan Diagnosis of Occult Groin Hernia." Hernia 16.3 (2011): 307-14. Print.
Roberts, James R., Catherine B. Custalow, Todd W. Thomsen, and Jerris R. Hedges. Roberts and Hedges' Clinical Procedures in Emergency Medicine. Print.
Solomon, Caren G., Robert J. Fitzgibbons, and R. Armour Forse. "Groin Hernias in Adults." New England Journal of Medicine N Engl J Med 372.8 (2015): 756-63. Print.
Strange, Chad D., Krista L. Birkemeier, Spencer T. Sincleair, and J. Robert Shepherd. "Atypical Abdominal Hernias in the Emergency Department: Acute and Non-acute." Emerg Radiol Emergency Radiology 16.2 (2008): 121-28. Print.
Derikx, Joep P.m., Dirk H.s.m. Schellekens, and Stefan Acosta. "Serological markers for human intestinal ischemia: A systematic review." Best Practice & Research Clinical Gastroenterology 31, no. 1 (2017): 69-74.
Adriana Segura Olson, MD. Assistant Professor, Assistant Program Director, Department of Emergency Medicine, University of Texas Health San Antonio
Hepatic Encephalopathy In The ED
Over 600,000 adults in the United States have liver cirrhosis, and as many as 30 to 45 percent of these patients will develop overt neuropsychiatric abnormalities. This week we discuss when patients present to the emergency department with signs suggestive of hepatic encephalopathy and how to best evaluate, manage and disposition this complex group.