The author underscores the central position the emergency provider may hold in identifying and supporting victims of trafficking
Molar Pregnancy
Written by: Conner Morton, MD (NUEM ‘26) Edited by: August Grace (NUEM ‘24)
Expert Commentary by: Dana Loke, MD, MS
Expert Commentary
Thank you to Drs. Morton and Grace for this excellent infographic highlighting molar pregnancy and its relevance to Emergency Medicine providers. While rarer than other pregnancy issues seen in the Emergency Department, molar pregnancy is an important diagnosis for Emergency Medicine providers to be able to recognize, understand, and treat. As listed in the above post, symptoms of molar pregnancy can be similar to viable pregnancy and its complications, so it is important to obtain a timely ultrasound in any patient with unknown pregnancy location or if considering molar pregnancy. While molar pregnancy is not a difficult diagnosis to make as long as an ultrasound is done, it is important to be wary of its complications and treat appropriately. This includes monitoring vitals, specifically in consideration of hemorrhage and pre-eclampsia, with resuscitation and transfusion as needed. Consultation with OB/GYN should occur in the Emergency Department for immediate next steps, which will include procedural management. When updating the patient about the diagnosis and next steps, make sure to sensitively explain that molar pregnancy is nonviable. Lastly, make sure to complete a comprehensive review of systems and physical exam; molar pregnancy is associated with choriocarcinoma, which is known to spread widely and aggressively throughout the body.
Dana Loke, MD, MS
Assistant Professor
Emergency Medicine
University of Wisconsin-Madison
How To Cite This Post:
[Peer-Reviewed, Web Publication] Morton, C. Grace, A. (2024, Apr 1). Molar Pregnancy. [NUEM Blog. Expert Commentary by Loke, D. Retrieved from http://www.nuemblog.com/blog/molar-pregnancy
Other Posts You May Enjoy
Resuscitative Hysterotomy
Written by: Aldo Gonzalez, MD (NUEM ‘23) Edited by: Justine Ko, MD (NUEM ‘21)
Expert Commentary by: Paul Trinquero, MD (NUEM '19) & Pietro Bortoletto, MD
Introduction
Resuscitative hysterotomy (RH) is the new term for what was previously called perimortem cesarean delivery (PMCD). The new nomenclature is being adopted to highlight the importance of the procedure to a successful resuscitation during maternal cardiopulmonary arrest (MCPA). It is defined as the procedure of delivering a fetus from a gravid mother through an incision in the abdomen during or after MCPA. The goal of the procedure is to improve the survival of the mother and the neonate.
Physiology
There are physiologic changes that occur during pregnancy which reduce the probability of return of spontaneous circulation (ROSC) during cardiac arrest. Physiologic anemia of pregnancy reduces the oxygen carrying capacity of blood and results in decreased delivery of oxygen during resuscitation. The large gravid uterus elevates the diaphragm and reduces the lung’s functional reserve capacity (FRC), which when combined with increased oxygen demand from the fetus results in decreased oxygen reserves and resultant risk for rapid oxygen desaturations. The size of a gravid uterus at 20 weeks results in aortocaval compression which reduces the amount of venous return from the inferior vena cava and reduces cardiac output during resuscitation. The theory behind resuscitative hysterotomy is to increase the probability of ROSC by reducing the impact of aortocaval compression.
Supporting Evidence
A 2012 systematic review primarily investigated the neonatal and maternal survival rates after perimortem cesarean delivery and secondarily attempted to evaluate maternal and fetal neurological outcome and the ability to perform the procedure within the recommended time frame.
Inclusion Criteria
original articles, case series, case reports and letters to the editor, and reports from databases
had minimum of least five clinical details of the case (e.g. patient age, gravidity, parity, obstetric history, medical history, presenting rhythm, or location of arrest)
AND
the care administered (chest compression, ventilation, monitoring, drug administration)
AND
maternal return of spontaneous circulation or survival to hospital discharge or fetal neonatal outcome
Exclusion Criteria
Post-delivery arrests
Studies without enough data to understand the details of the arrests
Studies with unclear maternal and fetal outcomes
Population
Pregnant woman that
(1) had a cardiac arrest or a non-perfusing rhythm
(2) received chest compression and/or advanced life support medications and/or defibrillation
Average maternal age: 30.5±6.5 years (median 32, range 17–44, IQR, 26.5–35.5, n = 80)
Gravidity: 2.5±1.5 (median 2, range 1–7, IQR 1–4, n = 59)
Parity: 1.1±1.3 (median 1, range 0–6, IQR 0–2, n = 57)
Singleton Pregnancies: 90.4% (n = 85)
Average gestational age at arrest: 33±7 weeks (median 35, range 10–42, IQR 31–39, n = 85)
Results
for cases undergoing PMCD, earlier time from arrest to delivery was associated with increased survival (p < 0.001, 95%CI 6.9–18.2)
surviving mothers: 27/57; 10.0±7.2 min (median 9, range 1–37)
non-surviving mother: 30/57; 22.6±13.3 min (median 20, range 4–60)]
for neonates delivered by PMCD/RH earlier time from arrest to delivery was associated with increased survival (p = 0.016)
surviving neonates: 14±11 min (median = 10, range = 1–47)
non-survivor neonates: 22±13 min (median = 20, range = 4–60)
Only 4 cases met the timeframe of less than minutes
Take-Aways: Performing a PMCD/RH in the 4-5 minutes time frame is difficult. However, PMCD/RH beyond the proposed time is still beneficial and earlier time to delivery from arrest is associated with better outcomes
Guideline Recommendations
Perform basic life support (BLS) in the same way as non-pregnant patients
Place patient in supine position
Left lateral decubitus (left lateral tilt) positioning is no longer recommended during compressions because of reduced efficacy of chest compressions
No modification of Chest compressions
Rate: 100-120 per minute
Depth: at least 2 inches (5 cm)
Allow for full chest recoil between compressions
Avoid interruptions as much as possible
No modification of Ventilation
Use bag-ventilation
Compression to breath ratio: 30:2 before advanced airway
Perform advanced cardiac life support (ACLS) as in non-pregnant women
No modification of Ventilation
Once breath every 6 seconds (10 BPM) with advanced airway
No modification of medications
Use 1 mg Epinephrine of epinephrine every 3-5 minutes
No modification to defibrillation
Use adhesive pads on patient
Place in anterolateral position
Lateral pad should be placed under breast tissue
Defibrillate for Ventricular fibrillation or Ventricular tachycardia
Use usual Voltages
Biphasic: 120-200 Joules
Resume compressions after shock is delivered
Special considerations during resuscitation
Obtain access above the diaphragm to minimize the effect of aortocaval compression on the administration of drugs
Perform left uterine deviation during resuscitation to reduce aortocaval compression
If a gravid patient suffers a cardiac arrest mobilize resources to prepare for the need for resuscitative hysterotomy and the resuscitation of the fetus early
Palpate the size of the gravid uterus
If above the height of the umbilicus then patient is most likely greater than 20 weeks gravid and a candidate for RH
Strongly consider performing RH (PMCD) if the patient does not achieve ROSC by the 4-minute mark and qualified staff to perform the procedure are present
Aim to have the procedure done by the 5-minute mark
Consider performing RH (PMCD) sooner if maternal prognosis is poor or prolonged period of pulselessness
RH should be performed at the site of the resuscitation
Do not delay procedure to prepare abdomen
May pour iodine solution over abdomen prior to incision
Do not delay procedure for surgical equipment if scalpel is available
Continue performing LUD while performing RH
Figure 1: One-handed left uterine deviation technique
Figure 2: Two-handed left uterine deviation technique
Steps for Resuscitative Hysterectomy
Pre-procedure
Gather supplies to perform RH
Personal Protective Equipment
Gloves
Face mask
Apron/gown
Resuscitative Hysterotomy Equipment
Scalpel(the minimum equipment to perform procedure)
Blunted Scissors
Clamps/Hemostats
Gauze
Suction
Large absorbable sutures
Needle Holder
Antiseptic Solution
Neonatal resuscitation equipment
Dry Linens
Neonatal Bag Valve Mask
Neonatal Airway supplies
Suction
Umbilical venous access equipment
Neonatal resuscitation drugs
Baby Warmer
Plastic Bag
Form teams to perform Resuscitative Hysterotomy
Resuscitative Team
Resuscitative Hysterotomy Team
Neonatal Resuscitation Team
Procedure
Maintain patient in supine position and continue compressions
Continue Left Uterine Deviation until the start of incision
Quickly prepare the skin with antiseptic solution (do not delay for skin prep)
Perform midline vertical Incision with scalpel on the abdomen from pubic symphysis to umbilicus and cut through skin and subcutaneous tissue until fascia is reached
Use fingers to bluntly dissect the rectus muscle fascia access the peritoneum (can use scalpel or blunt scissors)
Locate the uterus and differentiate it from the bladder (bladder yellow and enveloped in fatty tissue)
Make a vertical incision from the lower uterus to the fundus with scalpel (can use blunt scissors)
If the placenta is encountered while entering the uterus, cut through it
Use a cupped hand to locate the fetal part closest to pelvis
Elevate the located fetal part and pass through uterine incision while applying transabdominal pressure with other hand
Use traction and transabdominal pressure to deliver the rest of the baby
Clamp the cord at two spots and cut in between both clamps
Hand the baby to the neonatal team
Deliver placenta with gentle traction
Post-procedure
Continue performing compressions
Consider stopping if ROSC not achieved after several rounds and depending on the cause of PMCA
Give medications to promote uterine contraction
Analgesia and sedation may be required if patient achieves ROSC
Bleeding will be worse if ROSC achieved and may require pharmacologic and nonpharmacologic interventions
Closure will depend on whether the patient achieves ROSC and may necessitate careful closure to prevent further bleeding. Best performed by an obstetrician. If an obstetrician is unavailable, pack the uterus with gauze and clamps actively bleeding vessels to reduce bleeding.
Administer prophylactic antibiotics
References
Einav, S., et al. (2012). "Maternal cardiac arrest and perimortem caesarean delivery: evidence or expert-based?" Resuscitation 83(10): 1191-1200.
Jeejeebhoy, F. M., et al. (2015). "Cardiac Arrest in Pregnancy: A Scientific Statement From the American Heart Association." Circulation 132(18): 1747-1773.
Kikuchi, J. and S. Deering (2018). "Cardiac arrest in pregnancy." Semin Perinatol 42(1): 33-38.
Parry, R., et al. (2016). "Perimortem caesarean section." Emerg Med J 33(3): 224-229.
Rose, C. H., et al. (2015). "Challenging the 4- to 5-minute rule: from perimortem cesarean to resuscitative hysterotomy." Am J Obstet Gynecol 213(5): 653-656, 653 e651.
Soskin, P. N. and J. Yu (2019). "Resuscitation of the Pregnant Patient." Emerg Med Clin North Am 37(2): 351-363.
Walls, R. M., et al. (2018). Rosen's emergency medicine: concepts and clinical practice. Philadelphia, PA, Elsevier.
Expert Commentary
This is an excellent review of an extremely rare, but potentially life-saving procedure. It may seem daunting to perform (and it should), but the evidence would say that a resuscitative hysterotomy (RH), especially if performed promptly, drastically improves survival during the catastrophic scenario of maternal cardiac arrest. This is even more important because these patients are young (and often relatively healthy) and could potentially have decades of meaningful quality of life if they can survive the arrest. That being said, this procedure is so rare that most of us not only have never performed it, but often have never even seen it. Not only that, but unlike other rare lifesaving procedures (such as cricothyroidotomy or resuscitative thoracotomy), RH is extremely difficult to practice in cadaver labs due to the unavailability of pregnant cadavers. So, we are left with the next best thing: familiarizing ourselves with the anatomy, physiology, and simplified technique of the procedure and mentally rehearsing it so that when the time comes, we can be ready.
For these rare procedures, in addition to the excellent and thorough review above, it is also helpful to simplify and rehearse the fundamental steps. I’m not an obstetrician and certainly not an expert on this procedure, but I’ve mentally prepared myself for what I would do in the event that I am faced with this grave situation and categorized it into the following simplified five step plan. Also, prior to writing this commentary I got a curbside consult from a friend from med school and actual obstetrician and gynecologic surgeon, Dr. Pietro Bortoletto.
First off, the indications-- basically, a pregnant woman estimated to be >20 weeks EGA who has suffered a cardiac arrest. Don’t worry about the 4 minutes, make the decision to perform a RH right away and start prepping. Delegate someone to call the appropriate resuscitation teams if available. Then start the procedure.
Step 1: Setup. You probably don’t have a c section kit in your trauma bay, so instead open the thoracotomy tray and you’ll have most of what you need. Go ahead and set aside the finochietto rib spreaders so that you don’t have a panic attack trying to remember how to put those together with other people watching. But everything else you’ll need will be in that tray (basically a scalpel, blunt scissors, and hemostats).
Step 2: Cut into the Abdomen. Splash prep the abdomen with betadine. Then make your long vertical incision from the uterine fundus to the pubic symphysis. Cut through the skin and subcutaneous tissue then bluntly separate the rectus and enter the peritoneum with scalpel or blunt scissors. Extend the peritoneal incision with blunt scissors.
Step 3: (carefully) Cut into the Uterus. First, locate the uterus. Then, take a deep breath and remember that there is a fetus inside the uterus. With that terrifying thought in mind, cut vertically into the uterus, insert your fingers, and extend the incision upwards with blunt scissors and a steady hand. If you encounter an anterior placenta, cut right through it.
Step 4: Delivery. Deliver the fetus either by cupping the head and elevating it through the incision or by grabbing a leg, wiggling out the shoulders, and then flexing the head. Hand over the neonate to whoever is taking the lead on the neonatal resuscitation (will need to be warmed, stimulated, and potentially aggressively resuscitated). Clamp and cut the cord, leaving a long enough umbilical stump for an easy umbilical line if needed. Then using gentle traction, attempt delivery of the placenta. If it isn’t coming easily, leave it alone so as not to stir up more bleeding.
Step 5: Extra credit. If you’ve made it this far as an emergency physician and there is still no obstetrician in sight, you can continue resuscitation, focusing on stopping the uterine bleeding. While you don’t need to close the fascia or skin, it can be helpful to close the uterine incision to prevent additional blood loss. You can do this with a whip stitch using 0-0 vicryl (or if that seems like showing off, you can just pack it with sterile gauze. If you’ve got it handy, give 10 IU oxytocin to stimulate uterine contraction and further slow bleeding. Feel free to order some antibiotics as well. Otherwise, continue maternal resuscitation following typical ACLS.
The big picture here is that this is a heroic, potentially life-saving procedure that most of us will never do. But we can all take a few minutes to read an excellent review like the blog post above, watch a video, and mentally walk ourselves through the simplified steps. That preparation will afford us some much-needed confidence if we are ever faced with this terrifying scenario.
Paul Trinquero, MD
Medical Director
Department of Emergency Medicine
US Air Force Hospital - Langley
Pietro Bortoletto, MD
Clinical Fellow
Reproductive Endocrinology & Infertility
Weill Cornell Medical College
How To Cite This Post:
[Peer-Reviewed, Web Publication] Gonzalez, A. Ko, J. (2021, Dec 13). Resuscitative Hysterotomy. [NUEM Blog. Expert Commentary by Trinquero, P and Bortoletto, P]. Retrieved from http://www.nuemblog.com/blog/resuscitative-hysterotomy.
Other Posts You May Enjoy
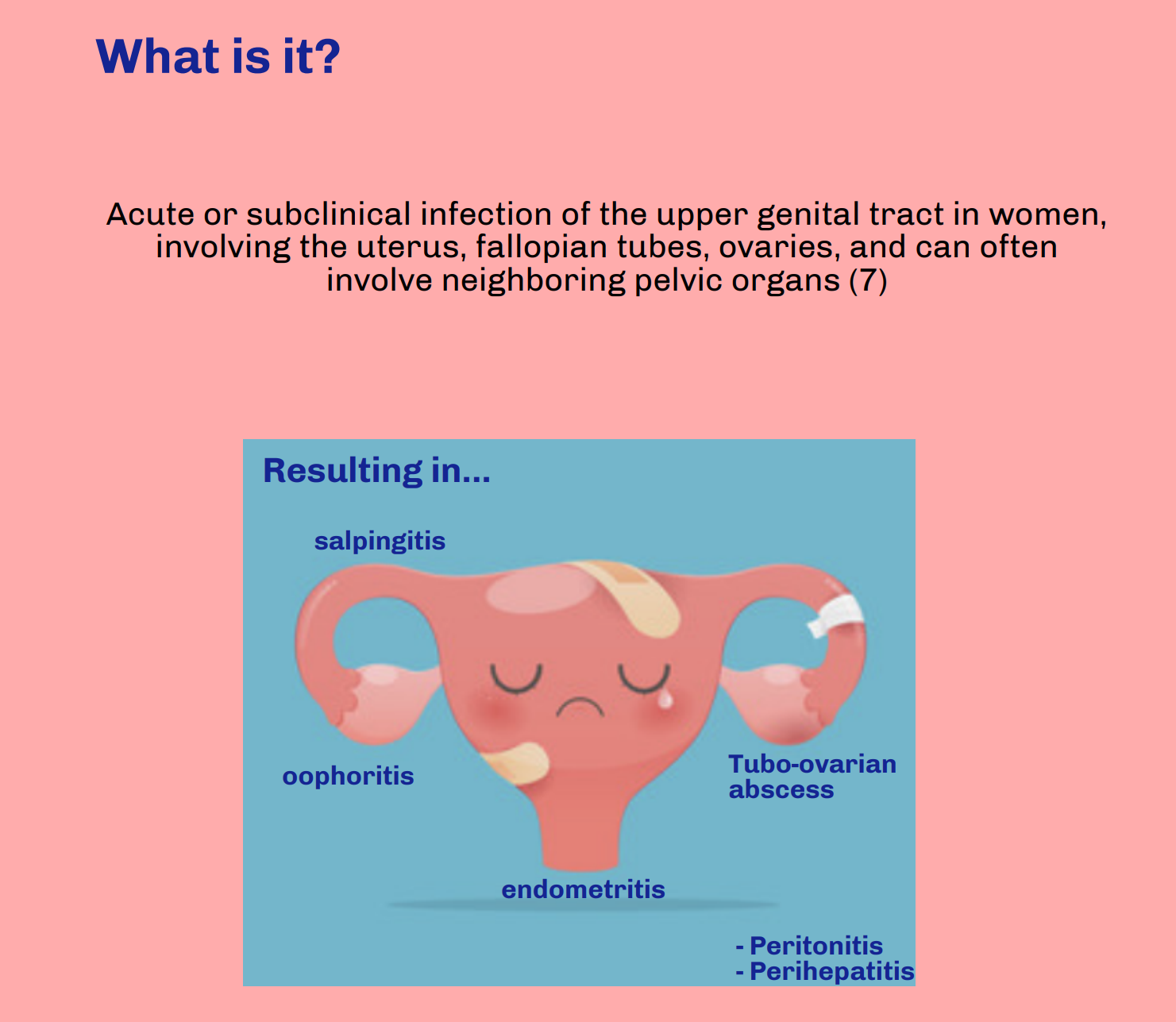
Pelvic Inflammatory Disease
Written by: Niki Patel, MD, MD (NUEM ‘22) Edited by: Luke Neill, MD (NUEM ‘20) Expert Commentary by: Gabby Ahlzadeh, MD
Expert Commentary
Thanks for this clear and succinct post. The differential diagnosis of lower abdominal and pelvic pain is extremely broad in both premenopausal and post-menopausal women. This is when the sexual history becomes important. A question we often overlook as part of the sexual history is asking about dyspareunia, which may help differentiate gynecological from intra-abdominal causes of abdominal pain, specifically in the case of PID.
Patients with PID are frequently misdiagnosed with a urinary tract infection because they may have urinary symptoms, but the urinalysis often shows sterile pyuria, which should raise your suspicion for PID.
And while the utility of the pelvic exam is constantly scrutinized and questioned in patients with vaginal bleeding, it is impossible to diagnose PID without it. Having said that, the clinical diagnosis is only 65-90% specific so even minimal symptoms with no other explanation warrant antibiotic therapy to reduce further complications.
Underdiagnosis is even more significant in the adolescent patient population, who are at highest risk for developing PID. Over 70% of PID diagnoses among adolescents are made in the ED, with approximately 200,000 adolescents diagnosed annually. If the patient is accompanied by a family member or friend, having them step out to better elicit a sexual history is essential. HIV and syphilis testing should also be considered while these patients are in the ED.
Ensuring follow-up for these patients within 48-72 hours is essential and must be emphasized. Patients should understand the complications of PID and the importance of antibiotic compliance prior to discharge, especially in younger patients.
Gabrielle Ahlzadeh, MD
Clinical Assistant Professor of Emergency Medicine
University of Southern California
How To Cite This Post:
[Peer-Reviewed, Web Publication] Patel, N. Neill, L. (2021, Apr 5). Pelvic Inflammatory Disease. [NUEM Blog. Expert Commentary by Ahlzadeh, G]. Retrieved from http://www.nuemblog.com/blog/pelvic-inflammatory-disease
Other Posts You May Enjoy
Vaginal Self Swabbing
Written by: Alex Herndon, MD (NUEM ‘21) Edited by: Vidya Eswaran, MD (NUEM ‘2020) Expert Commentary by: Matt Klein, MD, MPH
The Antiquated Pelvic Exam?
As Emergency Medicine physicians the demand to see more patients and expedite turnaround times all while providing quality care and maintaining patient satisfaction is steep. Our attempt to meet these standards is truly tried when posed with having to perform a pelvic exam during a busy shift. Introducing patient self-swabbing: an opportunity to provide quality care while saving precious time and resources.
Chlamydia is the most common infection reported in the United States, particularly among high risk populations such as females ages 15 to 24 [1]. These patients are most likely to present to the Emergency Department for their symptoms, thus it remains the task of the Emergency Medicine physician to screen and diagnose sexually transmitted infections to aid in avoiding complications of infection such as pelvic inflammatory disease, infertility, and ectopic pregnancy [1].
The gold standard for making the diagnosis has traditionally been through the physician-obtained endocervical swab on pelvic exam, an exam than has been shown to be physically and emotionally uncomfortable for a majority of patients, as well as low yield [2]. In 2012 a study at an urban sexual health center on women 16 years of age or older presenting with and without vaginal discharge or bleeding compared self-swabs to physician collected endocervical samples. Self-swabs were more sensitive in detecting chlamydia by nucleic acid amplification tests (NAAT) when compared to physician swabs; compared to self-swabs 1 out of 11 cases were missed by physician collected swabs, an overall 9% miss rate [3]. Patient self-swabs are equivalent to physician swabs in detecting gonorrhea [4] The difference in sensitivity has been attributed to patients having more contact time with the vaginal wall and removing more mucus when self-swabbing compared to when physicians collect samples [5].
Another study performed at two urban teaching hospitals in New York looked at self-swabs versus physician collected swabs in order to assess if self-swabs were as sensitive at diagnosing chlamydia infection within the Emergency Department. Overall self-swabs were 91% sensitive and 99% specific at identifying infection, thus deemed a reasonable alternative to physician collected samples, and implemented as an alternative in order to save time and resources.(6) Currently, self-swabbing is supported by ACOG, AAFP, and the CDC [2, 7, 8].
So why aren’t we doing this more? Inherently, as Emergency Medicine physicians, we are always seeking out the big, the bad, and the ugly, be it a fungating mass, or a case of pelvic inflammatory disease. While the idea of the self-swab shouldn’t obviate the pelvic exam, it can be useful in populations with a history and symptoms suggestive of a sexually transmitted infection, as well as for patients seeking screening after known exposure, or even for patients who refuse to undergo a pelvic exam [2, 7] In addition, patients prefer to obtain self-swabs.(8) While one can argue time is still lost in having to instruct the patient on how to self-swab, its practice in the outpatient clinical setting has become so common that there are numerous resources to aid in patient education, including easy-to-understand diagrams, like the one below, made to adorn bathroom walls [9].
Self swabs have been shown to be less messy, cost-effective, as well as thought to be easy to perform by the majority of patients [10]. When striving to increase efficiency, all the while improving patient care, every second counts. Self-swabbing is one method that can buy back time well-spent.
Expert Commentary
While this terrific post specifically focuses on the use of self-administered vaginal swabs for the evaluation of cervicitis in the emergency department, the broader utility of the pelvic exam for ED patients has been repeatedly called into question [1, 2]. As you correctly point out, pelvic exams are uncomfortable, can be distressing for patients, and frequently introduce delays in the patient’s care.
In addition to the evidence supporting self-swabs cited in this post, a 2018 ED-based study suggests the pelvic exam does not increase the sensitivity or specificity of diagnosing chlamydia, gonorrhea, or trichomonas when compared to taking a history alone [3]. While any individual study of this type will be limited by methodologic issues, there does appear to be broad support in the literature for routine use of self-administered swabs.
So why aren’t we doing this? Clinical practice can be slow to change, and that seems to be the case for this topic. I also think this highlights a fundamental feature of the emergency medicine mindset: the emphasis on identifying “bad” conditions, despite an anticipated low likelihood. While I have never personally visualized an unexpected cervical malignancy or traumatic injury during a pelvic exam in the ED, many emergency clinicians fear “missing something” in the absence of direct visualization. But as the 2018 paper highlights, taking an appropriate history should mitigate these concerns. Finally, as this post mentions, patients must be instructed on how to properly perform a self-administered swab, and any education should be appropriate to the patient’s primary language and degree of health literacy.
References
Close R, Sachs C, Dyne P. Reliability of bimanual pelvic examinations performed in emergency departments. West J Med. 2001;175(4):240-4.
Brown J, Aristizabal J, Fleming R, et al. Does pelvic exam in the emergency department add useful information. West J Emerg Med. 2011;12:208-212.
Farrukh S, Sivitz A, Onogul B, et al. The additive value of pelvic examinations to history in predicting sexually transmitted infections for young female patients with suspected cervicitis or pelvic inflammatory disease. Ann Emerg Med. 2018;72(6):703-712.
Dr. Matthew R Klein, MD, MPH
Assistant Professor of Emergency Medicine
Assistant Program Director
Department of Emergency Medicine
Northwestern Memorial Hospital
How To Cite This Post
[Peer-Reviewed, Web Publication] Herndon, A. Eswaran, V. (2020, Sep 7). Vaginal Self Swabbing. [NUEM Blog. Expert Commentary by Klein, M]. Retrieved from http://www.nuemblog.com/blog/vaginal-self-swabbing.
Other Posts You May Enjoy
References
Wiesenfeld H. Screening for Chlamydia trachomatis Infections in Women. New England Journal of Medicine. 2017; 376(22):2197-2198. doi:10.1056/nejmc1703640.
Smith R. The Unnecessary Pelvic Exam. Sinai-Grace Emergency Medicine Residency. http://emsgh.com/wp/the-unnecessary-pelvic-exam-dr-smith/.
Schoeman S, Stewart C, Booth R, Smith S, Wilcox M, Wilson J et al. Assessment of best single sample for finding chlamydia in women with and without symptoms: a diagnostic test study. BMJ 2012; 345:e8013
Stewart C, Schoeman S, Booth R, Smith S, Wilcox M, Wilson J et al. Assessment of self taken swabs versus clinician taken swab cultures for diagnosing gonorrhoea in women: single centre, diagnostic accuracy study. BMJ 2012; 345:e8107
Leon R. Indications and value of self-administered vaginal swabs for STIs and vaginitis. Faculty of Medicine: This Changed My Practice. November 2017. https://thischangedmypractice.com/self-administered-vaginal-swabs-sti-vaginitis/.
Berwald N, Cheng S, Augenbraun M, Abu-Lawi K, Lucchesi M, Zehtabchi S. Self-administered Vaginal Swabs Are a Feasible Alternative to Physician-assisted Cervical Swabs for Sexually Transmitted Infection Screening in the Emergency Department. Academic Emergency Medicine. 2009;16(4):360-363. doi:10.1111/j.1553-2712.2009.00359.x.
Lunny C, Taylor D, Hoang L, et al. Self-Collected versus Clinician-Collected Sampling for Chlamydia and Gonorrhea Screening: A Systemic Review and Meta-Analysis. Plos One. 2015;10(7). doi:10.1371/journal.pone.0132776.
Page C, Mounsey A, Rowland K. PURLs: Is self-swabbing for STIs a good idea?. J Fam Pract. 2013; 62(11):651-3.
Self-Collected Vaginal Swabs for Gonorrhea and Chlamydia. NC Sexually Transmitted Diseases Public Health Public Health Program Manual/Laboratory Testing and Standing Orders. 2011.
Fielder RL, Carey KB, Carey MP. Acceptability of Sexually Transmitted Infection Testing Using Self-collected Vaginal Swabs Among College Women. Journal of American College Health. 2013;61(1):46-53. doi:10.1080/07448481.2012.750610.
Chief Complaint: Sexual Assault
Written by: Logan Wedel, MD (NUEM PGY-3) Edited by: Jason Chodakowski, MD (NUEM ‘20) Expert Commentary by: Erin Lareau, MD
ED track board reads: 24 F *****, CC: SA
• Unfortunately, this is not an uncommon complaint we see in the ED
• Stay engaged, and prepare for a prolonged patient stay
Sexual Assault has reached Epidemic Proportions in the United States and Globally
19.3% of women and 1.7% of men are raped at some point in their lifetime
Of female rape victims, 78.2% have their first experience of rape before the age of 25 Recent data suggests estimated cost is $122,461 per rape victim .
In one study it was found that alcohol/substance abuse was involved in over 50% of cases.
Only a small proportion of victims present to the emergency department
When they do the we play a vital role in treating injuries, providing prophylaxis, and collecting evidence that can be used to apprehend the attacker
It is not our role to judge the validity of the patient's accusations, to identify the attacker, nor file a police report. The latter is at the patient's discretion.
The process will be time consuming but these patients deserve our full attention: minimize distractions, sit down, provide deep empathy, and give them the space to tell their full story.
Perhaps most importantly you must provide patients with what was violently take from them: a sense of control and safety.
Step 1: Obtain History
Time and Location
Exact details if able to remember
Identity of the Attacker (if known) Number of individuals
Possible identifying information
Specific Encounter Details - Use Patient Quotes Penetration (vaginal, anal, oral)
Ejaculation?
Condom use?
Use of other foreign bodies?
Licking, kissing, biting?
Post Assault Activities
Shower, urination, defecation?
Did they change their tampon, diaphragm, or clothing? Any oral intake or vomiting?
Patient's Medical History
HIV and Hepatitis B status and vaccination
Recent consensual sexual encounters
Step 2: Physical Exam
General Physical Exam
Immediate and acute interventions always take precedent
Pelvic and GU exam
Can be done with Evidence Collection Kit If patient consents
Detailed skin and soft tissue exam
Again can be conducted with Evidence Collection
Step 3: Medical Management
Always tend to trauma first
Primary and Secondary surveys
Workup traumatic injuries (XR / CT / FAST)
Baseline Labs and Blood draws
CBC, CMP, LFT's, UA, Urine Pregnancy, HIV
Offer Medical Advice and Inform patient of Risks, and Potential Prophylaxis Options
High Risk (By Prevalence) --> Empiric Treatment
Chlamydia: 528.8 per 100,000 -> Azithromycin 1g PO
Gonorrhea:171.9 per100,000 -> Ceftriaxone 250mg IM
Trichomonas: 3.1% -> Metronidazole 2g PO
Bacterial V.: 29.2% -> Metronidazole 2g PO
Lower Risk--> PEP options
HIV: 0.1% vaginal / 2.0% Anal ->
Emtricitabine/Tenofovir 200/300mg PO: 1 tab QD
Raltegravir 400mg PO: 1 tab BID
Hep B: <1% ->
Hep B vaccine Series: Now, 1-2m, 4-6m
Hepatitis C : < 1% -> No known prophylaxis
Syphilis: 9.5 cases per 100,000 ->
RPR test at 6wks, 3m, 6m
PenicillinG2.4millionUIM
Pregnancy Risks
Dependent on Ovulatory Cycle:
3 days before ovulation: 15%
1-2 days before ovulation: 30%
Day of ovulation: 12%
1-2 Days after Ovulation: 0%
Emergency Contraception
Only if Urine Pregnancy Test Negative
Levonorgestrel 1.5mg PO
Step 4: Evidence Collection Kit --Best if within 72 hours
Obtain patient consent
Verbal Consent to Contact "Rape Victim Advocate"
Signed Consent for Sexual Assault Evidence Kit
Police must be Contacted//However patient does not have to talk with authorities
Patient can also decide to refuse evidence collection at any time
AppropriateAttire
Gloves,Gown,HairRestraint
Collect Articles of Clothing
Patient undresses on a sheet, which is supplied in the kit
Anything worn at the time of assault
Underwear: worn at the time, or up to 72hrs after
Individual Articles of Clothing in Separate Areas
Place Individually in collection bags, sealed with evidence tape
Medical/ForensicDocumentation
ObtainedDuringOriginalPatientHistory
Key Aspects as Documented Above
DetailedPhysicalExam
Head to Toe Inspection and Palpation
Documentation of ANY Injuries--size, location, color, pattern
If Significant, Notify Police to Have Evidence Tech Obtain Photographs
Genital/AnalExam
Normal Speculum Examination, with Detailed Documentation
Note Discharge, Bleeding, Stains, Semen, Foreign Material, Trauma
Detailed Description of all Anatomy in Male/Female GU Area
Swab Genital / Anal area if Contact Occurred (Lubricate with Sterile Water)
Note: Do Not Collect G/C or BV Swabs, Unless Patient is 10 Days out or Having Symptoms
Offer Empiric Treatment
Collection Specimens
Oral Specimens (4 Total)
Swabs: Tongue, Gum Line, Recessed Areas
Head Hair Combings
From Different Areas of Head
Place Comb with the Hair into Paper Sheet
Fingernail Specimens
Wood Stick to Scrap under Nails
Miscellaneous Bite Marks / Stains
Swab Area, Label Accordingly
Patient Blood on Filter Paper
Obtains Drops of Blood for Filter Paper
Pubic Hair Combings
Comb out Hair onto Supplied Paper
Cut Hair if Matted
Genital / Anal Swab (4 total)
Swab External Genital/Anal Area--Sterile Water to Lubricate
Follow Up Appointments and Safety Assessment
Prior to ED Discharge
Write for 28 day supply for HIV PEP: Medications as above
Primary Care Physician
Arrange for close follow up with PMD, ideally within 1 week
Send referral if patient is without a primary care physician
OB-GYN
In order to monitor potential GU trauma
HPV / STD surveillance
Infectious Disease
Within 5 days in HIV PEP is started--Due to potential toxicity
Close monitoring of liver function
Repeat testing as below
On-Going Screening / Laboratory Work --Per ID / Primary Care
HIV: at 6 weeks, 3 months, 6 months
Hepatitis B: 2nd Vaccination at 3 months / 3rd at 6 months
Hepatitis C: at 3-6 months
Syphilis: at 6 weeks and 3 months
Safety Assessment
If at risk for being assaulted again, strongly encourage patient's file a police report although this remains the patient's choice
If potentially unsafe going home provide resources for shelters
If social work isn't involved yet get them involved
Expert Commentary
This is a great summary of current epidemiology and ED clinical practices surrounding the care of sexual assault patients. To reiterate and expand upon your synthesis:
• Sexual assault is an extremely common traumatic injury that is underreported to physicians.
• Sexual assault victims may have multiple traumatic injuries, acute psychiatric needs, and complex social needs. A multidisciplinary approach to their care is often helpful, and necessary to reduce further psychological stress associated with the emergency department exam experience after an assault.
• Recently, the US Department of Justice has published guidelines for training forensic examiners of sexual assault patients, including sexual assault nurse examiners (SANEs) and sexual assault forensic examiner (SAFE). These professionals are specially trained to provide care for sexual assault patients, and to perform the evidence collection. They are often also trained in forensic photography. SANEs typically manage the entirety of the patient encounter. This includes coordination of prophylactic medications and proper follow up. Illinois currently has a program to train all RNs on the sexual assault exam, and requires a SANE nurse to be available in the ED.
• There are additionally trained SANE/SAFE providers who also specialize in adolescent/pediatric sexual assault forensics. These providers should be called upon when available for all children suffering from sexual assault, as there is a higher risk for additional trauma surrounding the exam in these populations.
• Our job as physicians should therefore focus on:
Identifying and treating additional medical or traumatic injuries
Counseling patients on prophylactic medication - as indicated by the exposures which you noted above
Reviewing expectations and follow up
Collaborating with our SANE colleagues, volunteer rape victim advocates, pharmacists, police departments, and social workers.
And as always, we should provide compassion and symptom relief to these patients undergoing an overwhelming traumatic event.
References:
http://www.illinoisattorneygeneral.gov/victims/sane.html. Accessed 2/12/2020
https://www.justice.gov/ovw/page/file/1090006/download. Accessed 2/12/2020
Erin Lareau, MD
Assistant Professor of Emergency Medicine
Northwestern Medicine
How To Cite This Post
[Peer-Reviewed, Web Publication] Wedel L, Chodakowski J. (2020, June 29). Chief complaint: sexual assault [NUEM Blog. Expert Commentary by Lareau E]. Retrieved from http://www.nuemblog.com/blog/chief-complaint-sexual-assault
Other Posts You May Enjoy
Resources
Avegno, Jennifer, MD et al. "Violence:Recognition,Management,Prevention Sexual Assault Victims in the Emergency Department: Analysis by Demographic and Event Characteristics."
The Journal of Emergency Medicine, Vol. 37 No. 3. 2009, pp. 328-344“BacterialVaginosisStatistics." Center for Disease Control and Prevention U.S. Department of Health & Human Services.
Breiding, Matthew J, PhD eta l. "Morbidity and Mortality Weekly Report: Prevalence and Characteristics of Sexual Violence, Stalking, and Intimate Partner Violence Victimization." Center for Disease Control and Prevention, U.S. Department of Health & Human Services.
Chisholm, Christian A. MD, et al. "Intimate Partner Violence and Pregnancy: Epidemiology and Impact." American Journal of Obstetrics & Gynecology Vol 217. No. 2. 2017, pp 141-144.
HIV/AIDS:HIVRiskFactors. Center for Disease Control and Prevention, U.S. Department of Health & Human Services.
"Northwestern Memorial Hospital Department of Emergency Medicine Clinical Care Guideline: Sexual Assault." https://access.nmh.org/f5-w- 68747470733a2f2f6e6d692e6e6d682e6f7267$$/wcs/blob/1390883725624/clinical -care- guideline-sexual-assault.pdf.
"Preventing Sexual Violence." Center for Disease Control and Prevention, U.S. Department of Health & Human Services.
"Sexually Transmitted Disease Surveillance 2017: Chlamydia." Center for Disease Control and Prevention U.S. Department of Health & Human Services.
"Sexually Transmitted Disease Surveillance 2017: Gonorrhea." Center for Disease Control and Prevention U.S. Department of Health & Human Services.
"Sexually Transmitted Disease Surveillance 2017: Syphilis." Center for Disease Control and Prevention U.S. Department of Health & Human Services.
Sugg, Nancy MD, MPH. "IntimatePartnerViolence: Prevalence, Health Consequences, Intervention." Medical Clinics of North America, Vol.99, No. 3 2015, pp.629-649.
"Trichomoniasis Statistics." Center for Disease Control and Prevention, U.S. Department of Health & Human Services. https://www.cdc.gov/std/trichomonas/stats.htm
Assisted Reproductive Technology
Written by: Jesus Trevino, MD (NUEM ‘19) Edited by: Keith Hemmert, MD (NUEM ‘18) Expert Commentary by: Lia Bernardi, MD
In 2015, assisted reproductive technology (ART) resulted in 72,913 live births, comprising approximately 1.6% of all infants born in the US (CDC 2017). As ART is becoming increasingly common, it is important that emergency medicine providers are familiar with this treatment modality to appropriately diagnose and manage maternal complications. This article will review the components and complications of ART that may present in the Emergency Department with a focus on the ovarian hyperstimulation syndrome (OHSS).
ART encompasses therapies that address all causes of infertility, which span both the male factor (e.g., sperm motility) and female factor (e.g., mechanical, ovulatory). In vitro fertilization is a common treatment strategy that overcomes reproductive barriers and involves: 1) controlled ovarian hyperstimulation, 2) oocyte retrieval, 3) oocyte fertilization and 4) oocyte implantation. Below is an outline of these in vitro fertilization steps along with associated complications.
Controlled ovarian hyperstimulation
There are numerous protocols available to recruit ovarian follicles and they differ in the type and intensity of exogenous stimulation (Speroff). Protocols are graded in order of increasing intensity (and success rate):
Natural cycle (i.e., no exogenous stimulation)
Minimal (i.e., clomiphene citrate)
Mild (i.e., clomiphene citrate + low-dose exogenous gonadotropins)
Aggressive (i.e., high-dose gonadotropins +/- gonadotropin-releasing hormone agonist or antagonist).
The most life-threatening complication of these strategies is ovarian hyperstimulation is OHSS (Adams):
Incidence - 0.5-5% of ART cycles; 0.1-2% involve severe presentations (Weinerman).
Pathophysiology - increased capillary permeability leads to edema, ascites, pleural and pericardial effusions; this inflammatory state may result in renal failure, respiratory failure and/or thromboembolism.
Timing - typically occurs within a week of exogenous HCG administration or in the peri-implantation period due to increases in endogenous HCG.
Risk factors - < 35 years, low BMI, gonadotropin-releasing hormone and/or analogues, hyperstimulation of ovarian follicles (detected via pelvic US), elevated estradiol levels
Presenting symptoms & signs - abdominal distention, rapid weight gain, peripheral edema, dyspnea, pulmonary edema/effusions, oliguria; minimize pelvic exams to avert ovarian cyst rupture and hemorrhage
Labs - +/- hyponatremia, AKI, estradiol > 3000 pg/mL (typically not practical in ED evaluation)
Management -
Criteria for outpatient management: normal VS, renal and hepatic labs, ovaries < 5 cm
Criteria for inpatient management: ovaries > 5 cm, ascites; admission is required for serial exams and pain control
Criteria for ICU: palpable ovaries, pleural effusions, ARDS, oliguria, hypotension, AKI, hepatic dysfunction; these patients require fluid resuscitation, +/- therapeutic thora/paracentesis, likely termination of ART cycle
In addition, controlled ovarian hyperstimulation has an increased incidence of ovarian torsion - 0.08% without OHSS and 3% with OHSS (Weinerman).
Oocyte retrieval
This step is usually performed under conscious sedation with ultrasound-guided, transvaginal needle aspiration. Complications may include (incidence %):
Vaginal puncture site bleeding (8%, Speroff)
Intraperitoneal bleeding (0.04-0.07%, Speroff)
Bowel perforation (0.04%, Weinerman)
Infection (0.3-0.6%, Speroff). Half of infections may present as TOA within 1-6 weeks after retrieval.
Oocyte fertilization
The in vitro part - sperm meets egg. There are rarely major maternal complications associated with this treatment step that present in the ED.
Oocyte implantation
Implantation occurs via a transcervical catheter under transabdominal US-guidance. As the success rate for single fertilized embryos is 10-25%, multiple embryos are implanted to increase implantation yield (Adams). Complications may include:
Multigestational pregnancy - 31-41% of IVF infants develop from multigestational pregnancies (Adams)
Ectopic pregnancy (0.7-4%, Speroff, Adams)
Heterotopic pregnancy (0.2-1%, Adams, Weinerman)
Lastly, patients with ART-facilitated pregnancies are at risk of thromboembolism (0.04-0.2%), especially in the presence of OHSS (4%, Weinerman).
Expert Commentary
This is an important review of complications that may arise in patients undergoing in vitro fertilization (IVF). Although patients who are planning for IVF can be assured that it is a generally safe process, there are medical issues that can occur throughout: during the ovarian stimulation phase, as a result of the oocyte retrieval or embryo transfer, or after a pregnancy is confirmed.
During the ovarian stimulation phase of the process, few medical complications typically arise. The most common reason that a patient would seek emergency care would be for ovarian torsion. Given that ovarian size increases significantly, any patient who presents with severe pain during stimulation should be assessed for this.
The most likely time a patient undergoing IVF would present to the ED would be following the oocyte retrieval. Complications can take place after the conclusion of stimulation or due to issues from the retrieval itself. One of the main issues that patients present for is ovarian hyperstimulation (OHSS). The most common time for this to happen is shortly after the oocyte retrieval, but patients may present in early pregnancy as well given that a rising hCG level worsens the syndrome. Evaluation and management of OHSS is reviewed succinctly above. Complications may also arise from the oocyte retrieval. Transvaginal ultrasound guided aspiration of ovarian follicles is performed using a needle that passes through the vagina. Possible complications include bleeding, infection, and/or or injury to other organs. Given that the needle is entering the vagina and the ovaries, bleeding can occur from the vagina or within the abdomen. Vaginal bleeding is typically quickly recognized and corrected prior to completing the procedure. Intraabdominal bleeding can be more difficult to identify and can potentially worsen after the patient is discharged. Patients with intraabdominal bleeding may present to the ED with symptoms of pain or hypotension. Imaging is generally helpful as part of the evaluation, but it is important to know that some bleeding generally occurs post-procedure even in an uncomplicated oocyte retrieval. Therefore imaging may reveal free fluid in a patient who does not have clinically significant ongoing bleeding. It is also important to be aware that if a patient has intraabdominal bleeding, surgical intervention is not always required. Given the complexities of surgical exploration in these patients, the goal is to expectantly manage those with intraabdominal bleeding unless surgery is absolutely necessary. In addition to bleeding, infection, ovarian torsion and cyst rupture can also occur following oocyte retrieval. Finally, some patients will have extreme constipation due to the IVF process and may present with abdominal pain after oocyte retrieval as a result.
Embryo transfers are a generally safe and low risk procedure. The procedure involves insertion of a sterile, soft catheter into the uterine cavity under ultrasound guidance with subsequent embryo release. Given the low risk nature of the procedure, complications after embryo transfer are very rare. Infection is theoretically possible, but unlikely.
The final IVF related complications to consider are those that occur in pregnancy. As ectopic pregnancies are possible after IVF, any pregnant woman who presents with symptoms concerning for an extrauterine pregnancy should be evaluated accordingly. Heterotopic pregnancies can also occur and should remain on the differential diagnosis if a woman has concerning pain after an intrauterine pregnancy is confirmed. Given that a woman’s ovaries remain enlarged after ovarian stimulation if a pregnancy is achieved, ovarian torsion should also be considered in patients with abdominal pain. Another complication that can develop at any point during the IVF treatment process, including during pregnancy, are VTEs. Given supraphysiolgic estrogen levels that occur due to ovarian stimulation, providers must bear in mind that this complication can arise.
Efficient recognition and treatment of the complications that can result from the IVF process are more likely when providers are well educated. Hopefully this review will improve the ability for patients undergoing IVF to be evaluated and treated most effectively when they present to the ED.
Lia Bernardi, MD
Assistant Professor
Department of Obstetrics and Gynecology
Feinberg School of Medicine
How to Cite This Post
[Peer-Reviewed, Web Publication] Trevino, J, Hemmert, K. (2020, May 11). Assisted Reproductive Technology. [NUEM Blog. Expert Commentary by Bernardi, L]. Retrieved from https://www.nuemblog.com/blog/assisted-reproductive-tech
References
Yang-Kauh C. Complications of gynecologic procedures, abortion, and assisted reproductive technology. Chapter 125. Emergency Medicine, Ed 2, 2013.
Speroff L, Fritz MA. Assisted Reproductive Technologies. Chapter 32. Clinical Gynecologic Endocrinology and Infertility, Ed 8, 2011.
Weinerman R, Grifo J. Consequences of superovulation and ART procedures. Semin Reprod Med. 2012 Apr;30(2):77-83.
ART Success Rates [Internet]. Centers for Disease Control and Prevention. Centers for Disease Control and Prevention; 2017 [cited 2017May2]. Available from: https://www.cdc.gov/art/artdata/index.html
A Practical Approach to Abdominal Imaging
Written by: Zach Schmitz MD (PGY-3) Edited by: David Kaltman, MD (PGY-4) Expert commentary by: Samir Abboud, MD
I often find myself in a gray zone when it comes to imaging abdominal pain. Any third year medical student worth their salt can tell you to get the RUQ ultrasound for the fat, fertile, forty year-old female with RUQ abdominal pain, fever, positive Murphy’s sign, and leukocytosis. However, my patients don’t usually fit the textbook, and I’m often thinking about what I might miss or see with test X vs test Y. Below, I’ll touch on a few common dilemmas where the optimal choice of imaging modality isn’t immediately clear by focusing on what you actually gain or lose by ordering one imaging test over another.
Scenario 1: Stone or Appendicitis?
Case: 62 year old female with HTN and HLD presents with RLQ pain. The pain woke her this morning and has been intermittent all day, occurring exclusively when she urinates. It is sharp, non-radiating, and increasing in intensity. She never had a pain like this and can now barely sit still. She has thrown up a few times over the past few hours. Vitals are stable and she is afebrile. She appears uncomfortable with RLQ tenderness but no rebound or guarding. Labs show slight leukocytosis, and urine has no blood.
If I suspect stone over appendicitis, will a CT without contrast miss appendicitis?
CT, MR, and US are well studied in their ability to detect and accurately diagnose appendicitis.[1]
CT with IV contrast is 96-100% sensitive and 91-100% specific. Per the American College of Radiology’s (ACR) appropriateness system, this is the most appropriate initial test for suspected appendicitis in adults.[2]
MR is 96% sensitive and 96% specific.[3]
Ultrasound has a wide range of data, with sensitivity ranging from 21-95.7% and specificity of 71-97%.[2]
CT without oral or IV contrast is nearly as useful for diagnosing appendicitis
A meta-analysis by Xiong et al included seven original studies investigating a total of 845 patients.[4]
Pooled sensitivity - 0.90 (95% CI: 0.86-0.92)
Pooled specificity - 0.94 (95% CI: 0.92-0.97)
Pooled positive likelihood ratio - 12.90 (95% CI: 4.80-34.67)
Pooled negative likelihood ratio - 0.09 (95% CI: 0.04-0.20)
Will a contrast enhanced CT for appendicitis ruin my chance to catch a kidney stone?
Non-contrast CT is the emergency standard in diagnosing nephrolithiasis with good reason - it is 97% sensitive and 95% specific.[5]
Will contrast ruin the ability to detect a stone?
This makes theoretical sense as stones and contrast are both hyper-intense on CT.
Sensitivity is decreased for small stones with contrast enhanced studies.
However, for stones > 3mm, sensitivity remains 95%.[5]
Only about 5% of stones that small ultimately require intervention.
Takeaways: You sacrifice a bit with a non-contrast study looking for appendicitis and a contrast enhanced study looking for stone, but both still work well. The American Urology Association recommends consultation for stones > 10mm.[6] Urology would also need to be involved with signs of sepsis, abscess, deterioration in renal function, intractable symptoms, or a transplant/solitary kidney. It seems I am very likely to see a stone requiring something other than watchful waiting on a CT with contrast. It is worse to miss an appendicitis than a 2mm stone, so contrast might make more sense if it’s close.
Scenario 2: RUQ Ultrasound after Negative CT San
Case: 84 year old male with a history of prostate cancer and hypertension presents from a nursing home with 4 days of diffuse abdominal pain. He has had no vomiting or bowel movements over this time. No urinary symptoms. He is hemodynamically stable, and his abdomen is diffusely tender (maybe worse in the RUQ) and distended but overall not terribly impressive. You order a CT for possible obstruction and it just shows a large stool burden. The gallbladder was visualized and looked normal.
If a CT is negative, should I get a RUQ US to look for cholecystitis?
RUQ Ultrasound
Per ACR, this is the most appropriate initial study for RUQ pain and suspected biliary disease.[7]
A 2012 meta analysis showed a sensitivity of 81% (95% CI 75-87%) and specificity of 88% for acute cholecystitis.[8]
It has the advantage of being dynamic, with a sonographic Murphy sign independently showing an 86% sensitivity and 35% specificity, positive predictive value of 43%, and negative predictive value of 82%.[9]
Computed Tomography (CT)
The same 2012 meta analysis only had one study with CT, but noted a sensitivity of 94% with fairly broad confidence intervals (95% CI 73-99) and a specificity of only 59%.[8]
ACR notes CT’s NPV for acute cholecystitis approaches 90%.[7]
A 2015 study looked at 101 patients who went to the OR and got both a CT and US. For acute cholecystitis, the sensitivities for CT and US were 92% and 79% respectively. For cholilithiasis, sensitivities for CT and US were 60% and 89% respectively.[10,11]
ACR states it is “usually appropriate” to proceed with CT for RUQ pain and suspected biliary disease with a negative or equivocal ultrasound.[7]
Although it lacks a sonographic murphy’s sign equivalent, its advantage is to help in operative planning and seeing complications, such as perforation or gangrene.
MRI has a sensitivity of 85% and a specificity of 81%. It is also considered “usually appropriate” by ACR if ultrasound is negative or equivocal[7]
Cholescintigraphy is the best imaging, showing 97% sensitivity and 90% specificity for acute cholecystitis. It is also the most appropriate study if you suspect acalculous cholecystitis.[7]
Takeaways: There are a few interesting points from this set of data. First, CT seems to have at least as good of ability to pick up cholecystitis compared to ultrasound. However, it is much worse in detecting gallstones themselves, which may be very relevant to a patient with abdominal pain. Second, the sensitivity of both RUQUS or CT isn’t really that great and we are probably missing a few episodes of cholecystitis. If there is a very high index of suspicion but negative imaging, it may be worthwhile to pursue additional workup. Overall, if the CT shows a normal gallbladder, and you are not worried about intractable biliary colic, the ultrasound probably won’t add much.
Scenario 3: Female Pelvic Pain
Case: 33 year old female with a history of chlamydia infection presenting with right sided abdominal pain. The pain has gradually been getting worse for 1 day. She has had a few episodes of vomiting. There is some white vaginal discharge she always has. On exam, she is tachycardic, normotensive, and febrile to 101.5. She has RLQ tenderness with voluntary guarding. On pelvic exam, there is some white vaginal discharge, CMT, R adnexal tenderness that seems less intense than her RLQ tenderness, and no masses noted.
If this patient had a normal appendix and ovaries after a contrast enhanced CT for appendicitis, how useful is an additional transvaginal ultrasound to rule out gynecologic pathologies?
For ovarian torsion:
A retrospective study of 834 patients showed the NPV of a contrast enhanced CT of the pelvis for ovarian torsion is 100%.[12]
A prospective study of 199 patients showed doppler ultrasound has a sensitivity and specificity for torsion of 100 and 97%.[13]
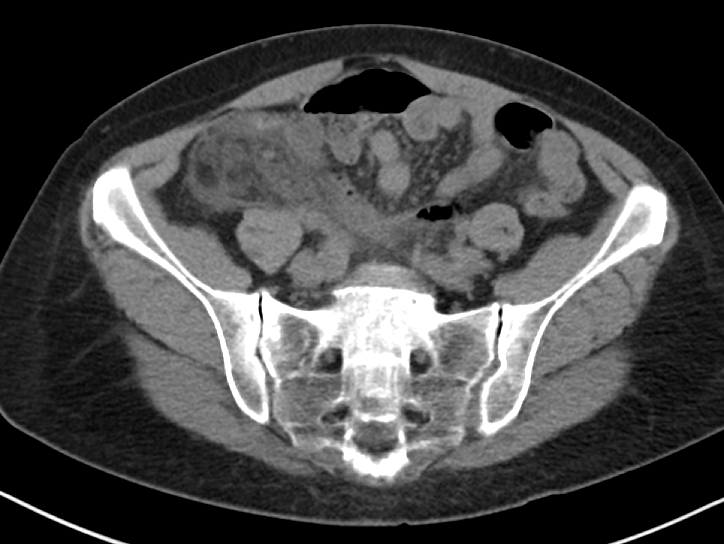
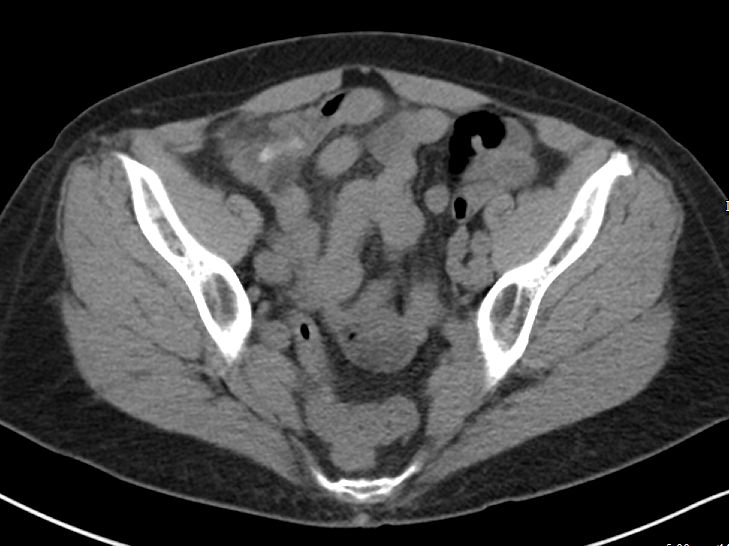
For Tubo-Ovarian Abscess (TOA):
CT is thought to be between 78 and 100% sensitive.[14]
2011 literature review gives a broad range of sensitivity and specificity for US in TOA with a sensitivity of 56-93% and specificity from 89-98%.[15]
Takeaways: ACR appropriates rates ultrasound as the most appropriate test for female pelvic pain.[14] However, it also rates CT with contrast as more appropriate for suspected appendicitis.[2] This patient raises concerns for both, and a CT was done first. CT is good for finding intra abdominal and pelvic abscess. It is more difficult to assess how useful ultrasound is for TOA, as many studies in the literature review were either before year 2000 or used a transabdominal approach. Overall, if someone has a CT scan for appendicitis that shows normal ovaries, the transvaginal ultrasound seems to add little for either torsion or TOA.
One potential dangerous conclusion from this set of data is that we should just CT everyone up front. While CT shows good sensitivities for many of the pathologies in question, simply ordering a CT first ignores the many good reasons - such as cost, radiation dose, speed, improved specificity and comparable sensitivity, resource utilization, sonographic murphy sign - RUQUS and pelvic ultrasound are the most appropriate initial tests for suspect biliary and pelvic pathology. That said, it a patient has an entirely normal CT that was already performed for other indicated reasons, the use of additional imaging may be unnecessary and should be considered carefully. Overall, the question of exactly what imaging test to order when ruling out common, emergent, abdominal pathologies is often a difficult one with shades of gray. By having a better understanding of exactly what type of information we are getting and missing from each test we order, emergency physicians can more quickly, safely, and accurately diagnose and treat our patients.
Expert Commentary
This is a thoughtful, well-reasoned approach to optimizing the imaging strategy in challenging, atypical clinical scenarios. To add a few nuances to some of the points raised:
When considering a contrast-enhanced versus non-contrast CT (both IV and PO) in the clinically ambiguous scenario, it is important to consider your patient’s body habitus. Figure 1 includes representative images from a non-contrast enhanced CT of a patient with a BMI above 25. You can clearly see the inflammatory stranding in the right lower quadrant mesenteric fat (Figure 1a) and portions of an appendicolith (Figure 1 b), in this patient who ultimately proved to have appendicitis. The natural contrast provided by the patient’s mesenteric fat in this scenario helps us work around the absence of IV contrast.
Figure 1a
Figure 1b
Figure 2 includes representative images from a contrast enhanced CT of a very thin patient, with a relative paucity of intra-abdominal fat. In this patient, the relative absence of natural contrast would greatly reduce our chances to diagnose appendicitis (or even identify the appendix) in the absence of IV contrast. PO contrast is additionally likely to be most helpful in very thin patients [Alabousi 2015].
Figure 2
The author asks (and answers) a very insightful question with regards to identifying kidney stones on contrast enhanced CT. A few points to add:
Assuming the contrast enhanced study is obtained prior to the excretory phase of imaging (and most routine studies are) ureteral stones should still be largely visible - the stones that will generally be more difficult to identify will be the non-obstructing stones still within the collecting system. Additionally, while there is indeed a small sacrifice in sensitivity for small stones with contrast enhanced studies, the identification of secondary complications is much improved.
Consider Figure 3, which demonstrates a 2 mm stone in the proximal left ureter identified on a contrast enhanced study. Notice the slightly delayed nephrogram on the left relative to the right, which could indicate a component of obstructive uropathy. Similarly, identification of such complications as pyonephrosis, pyelonephritis, and perinephric abscess is much improved with contrast enhanced images. For this reason, I would suggest that in the clinically ambiguous scenario, erring on the side of the contrast enhanced study would be wise.
Figure 3
It is important to note that the CT scanner installed in our emergency department is a dual-energy machine. Many of our other departmental scanners are dual-energy as well. With these scanners, we are able to apply algorithms to deconstruct the elemental composition of stones and provide more information than simply size and location - i.e. uric acid or non-uric acid stone - if requested. We can additionally generate virtual non-contrast images from the contrast-enhanced images, without exposing our patients to additional radiation. While it is tempting to think that we could recapture some of the sensitivity for renal stones using these virtual non-contrast images, this has unfortunately not been borne out in the literature at this time [Vrtiska 2010], though remains an area of continued investigation as imaging technology is further improved.
The advantages of dual-energy imaging are not only limited to the kidneys. With regards to the evaluation of biliary colic, virtual monochromatic images can be generated with resulting increased conspicuity of gallstones, even those that appear isodense to bile on the conventional images [Ratanaprasatporn 2018].
In general, if you find yourself with a high degree of suspicion for any disease process and discordant imaging findings, I would encourage you to call your radiologist. The additional clinical information exchanged during such a call may direct what additional data sets should be generated and what additional imaging studies may be of most benefit. Last, but certainly not least, that “second look” armed with additional clinical information can pick up on subtle findings that are, in isolation, entirely non-specific, but in a certain clinical scenario could clinch the diagnosis you are seeking.
References:
Alabousi A et al. Is Oral Contrast Necessary for Multidetector Computed Tomography Imaging of Patients With Acute Abdominal Pain? Canadian Association of Radiologists Journal. 2015;66(4): 318 - 322
Ratanaprasatporn L et al. Multimodality Imaging, including Dual-Energy CT, in the Evaluation of Gallbladder Disease. Radiographics 2018;38(1): 75-89
Vrtiska TJ et al. Genitourinary Applications of Dual-Energy CT. American Journal of Roentgenology. 2010;194: 1434-1442.
Samir Abboud, MD
Assistant Professor of Radiology
Northwestern University
How To Cite This Post:
[Peer-Reviewed, Web Publication] Schmitz, Z. Kaltman, D. (2020, Feb 10). An Approach to Abdominal Imaging. [NUEM Blog. Expert Commentary by Abboud, S]. Retrieved from http://www.nuemblog.com/blog/abdominal-imaging.
Other Posts You Might Enjoy…
References
Dahabreh IJ, Adam GP, Halladay CW, Steele DW, Daiello LA, Weiland LS, Zgodic A, Smith BT, Herliczek TW, Shah N, Trikalinos TA. Diagnosis of Right Lower Quadrant Pain and Suspected Acute Appendicitis. Comparative Effectiveness Review No. 157. (Prepared by the Brown Evidence-based Practice Center under Contract No. 290-2012-00012-I.) AHRQ Publication No. 15(16)-EHC025-EF. Rockville, MD: Agency for Healthcare Research and Quality; December 2015. www.effectivehealthcare.ahrq.gov/reports/final.cfm.
American College of Radiology. ACR Appropriateness Criteria®: RLQ pain. Available at https://acsearch.acr.org/docs/69357/Narrative/ Accessed 5/10/19.
Duke E, Kalb B, Arif-Tiwari H, et al. A Systematic Review and Meta-Analysis of Diagnostic Performance of MRI for Evaluation of Acute Appendicitis. AJR Am J Roentgenol 2016;206:508-17.
Xiong B, Zhong B, Li Z, Zhou F, Hu R, Feng Z, Xu S, Chen F. Diagnostic Accuracy of Noncontrast CT in Detecting Acute Appendicitis: A Meta-analysis of Prospective Studies. Am Surg. 2015 Jun;81(6):626-9.
Curhan G, Aronson M, Preminger G. Diagnosis and acute management of suspected nephrolithiasis in adults. UpToDate.com. April 30 2019.
Assimos D, Krambek A, Miller N et al. Surgical Management of Stones: AUA/Endourology Society Guideline (2016). https://www.auanet.org/guidelines/kidney-stones-surgical-management-guideline. Accessed 5/10/19.
American College of Radiology. ACR Appropriateness Criteria®: RUQ pain. Available at https://acsearch.acr.org/docs/69474/Narrative/ .
Kiewiet J.J., Leeuwenburgh M.M., Bipat S., et al: A systematic review and meta-analysis of diagnostic performance of imaging in acute cholecystitis. Radiology 2012; 264: pp. 708-720.
Bree, Robert L. Further observations on the usefulness of the sonographic Murphy sign in the evaluation of suspected acute cholecystitis. Journal of Clinical Ultrasound. March/April 1995.
Wertz JR1,2, Lopez JM3, Olson D4, Thompson WM1,2. Comparing the Diagnostic Accuracy of Ultrasound and CT in Evaluating Acute Cholecystitis. AJR Am J Roentgenol. 2018 Aug;211(2):W92-W97. doi: 10.2214/AJR.17.18884. Epub 2018 Apr 27.
Fagenholz, P et al. Computed Tomography Is More Sensitive than Ultrasound for the Diagnosis of Acute Cholecystitis. Surg Infect (Larchmt). 2015 Oct;16(5):509-12. doi: 10.1089/sur.2015.102. Epub 2015 Sep 16.
Lam A1, Nayyar M2, Helmy M2, Houshyar R2, Marfori W2, Lall C2.Assessing the clinical utility of color Doppler ultrasound for ovarian torsion in the setting of a negative contrast-enhanced CT scan of the abdomen and pelvis. Abdom Imaging. 2015 Oct;40(8):3206-13. Doi: 10.1007/s00261-015-0535-4.
Laufer, M. Ovarian and fallopian tube torsion. UpToDate. April 30 2019. https://www.uptodate.com/contents/ovarian-and-fallopian-tube-torsion?search=ovarian%20torsion&source=search_result&selectedTitle=1~70&usage_type=default&display_rank=1 .
Beigi, R. Epidemiology, clinical manifestations, and diagnosis of tubo-ovarian abscess. UpToDate. April 30 2019. https://www.uptodate.com/contents/epidemiology-clinical-manifestations-and-diagnosis-of-tubo-ovarian-abscess?search=tuboovarian%20abscess&source=search_result&selectedTitle=2~24&usage_type=default&display_rank=2 .
Lee DC1, Swaminathan AK. Sensitivity of ultrasound for the diagnosis of tubo-ovarian abscess: a case report and literature review. J Emerg Med. 2011 Feb;40(2):170-5. doi: 10.1016/j.jemermed.2010.02.033. Epub 2010 May 13.
American College of Radiology. ACR Appropriateness Criteria®: Female Pelvic Pain. Available at https://acsearch.acr.org/docs/69503/Narrative/ .
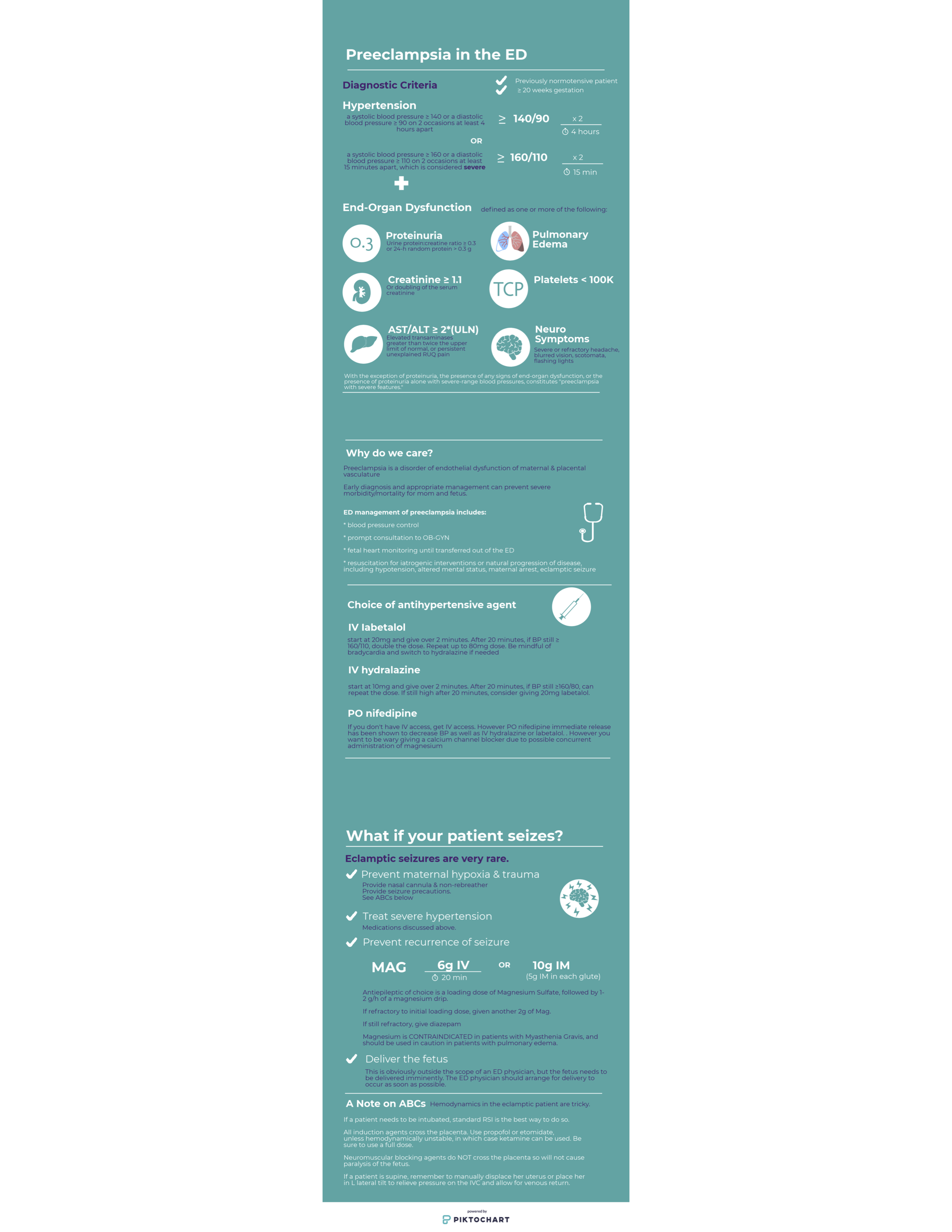
Preeclampsia
Written by: Priyanka Sista, MD (NUEM PGY-4) Edited by: Matt Klein, MD (NUEM ‘18) Expert commentary by: Shannon Lovett, MD
Expert Commentary
Thank you for this succinct guide to the diagnosis and management of preeclampsia in the ED.
“The eyes do not see what the mind does not know…..”. The biggest pitfall in the management of preeclampsia in the ED, is failing to consider and recognize the diagnosis. Recognition and prompt treatment of preeclampsia in the ED setting can be challenging due to the variety of presenting complaints. It is important to note that preeclampsia may occur anytime from 20 weeks gestation up to 6 weeks postpartum.
Postpartum preeclampsia tends to be more diagnostically challenging and depending on your facility, these patients are more likely to present to the ED than pregnant patients who often present to their obstetrician or to labor and delivery. Preeclampsia in the postpartum period most frequently occurs in the first 48 hours after delivery, but should be considered up to 6 weeks postpartum. Patients with postpartum preeclampsia often do not have hypertensive disease or preeclampsia during pregnancy.
The complaints associated with preeclampsia may be broad and vague- including but not limited to: headache, vision changes, swelling or rapid weight gain, nausea and vomiting, shortness of breath, and abdominal pain. Consider preeclampsia or eclampsia in the critical female patient that arrives in the ED with little known history- for example actively seizing, or in respiratory distress with pulmonary edema.
The treatment of preeclampsia can be broken down into three parts: treating the hypertension, reducing the risk or recurrence of seizures, and delivery of the fetus and the placenta. In the ED- our focus is on the first two, and involving our obstetric colleagues immediately. Blood pressure is most commonly treated with labetolol or hydralazine IV in the ED, and Mag should be given immediately for seizure prophylaxis (or to reduce recurrence of seizures in eclampsia).
Lastly, our obstetric and gynecology colleagues at ACOG have recognized the frequency that postpartum patients present to the ED, and have created this ED checklist that can be used as a reference for the management of postpartum preeclampsia- https://www.acog.org/-/media/Districts/District-II/Public/SMI/v2/19sm03a170703PPPreeclamCheckED1.pdf?dmc=1&ts=20190327T1949153065. Preeclampsia is a syndrome with potentially devastating consequences to mother and baby, and our early recognition and treatment can improve outcomes.
Shannon Lovett, MD
Associate Professor
Loyola University Medical Center
How To Cite This Post
[Peer-Reviewed, Web Publication] Sista P, Klein M. (2019, Sept 23). Preeclampsia. [NUEM Blog. Expert Commentary by Lovett S]. Retrieved from http://www.nuemblog.com/blog/preeclampsia.
Other Posts You May Enjoy
Post Partum Hemorrhage in the ED
Written by: Spenser Lang, MD (NUEM PGY-4) Edited by: Michael Macias, MD, (NUEM Graduate 2017) Expert commentary by: Annie Dude, MD
Introduction
A 26-year-old female G3P3 arrives via ambulance with heavy vaginal bleeding after having a precipitous home delivery of her third child. EMS reports a “pool of blood,” and en route to your facility she continued to bleed briskly.
Post partum hemorrhage (PPH) is a common and dangerous complication of child birth. According to CDC estimates, hemorrhage is the most common cause of maternal death in both developed and developing countries. About 2 out of every 100 births occur either at home, pre-arrival to the hospital or in the ED. With a trend towards home births and free standing delivery centers increasing dramatically in recent years, emergency physicians need to be able to recognize and treat this life threat.
The official diagnosis of PPH is volume-based, however this information is not easily obtainable in the emergency department. A more reasonable approach is to treat the PPH patient the same way you would a traumatic hemorrhage. Allow the patient’s vitals and visualized hemorrhaging to guide the aggressiveness of your resuscitation. Remember, a pregnant woman has ~40% extra circulating blood volume and can cope with a higher amount of blood loss than her non-pregnant counterpart.
Following an algorithmic approach as detailed below is essential to management of these patients.
Management
1. Notify an obstetrician.
- Having the obstetrician on board early will allow for mobilization of definitive treatment such as trans-arterial embolization and/or laparotomy should physical maneuvers, tamponade & uterotonics fail
2. Resuscitate.
- As with any resuscitation, begin with the simple ABC algorithm, addressing any issues as they are identified. Patients should be placed on 15 L of oxygen via face mask regardless of their saturation (if hemorrhage is significant, this will dramatically increase their blood oxygen levels via dissolved O2). If a patient is hemodynamically unstable, early administration of blood products should be considered over large volume crystalloid.
3. Obtain adequate access.
- Two large bore IVs will be necessary if aggressive resuscitation is needed. Consider an intraosseous line early if difficulty obtaining access. Send type and screen, CBC, coagulation panel and fibrinogen. Keep coagulopathy on your differential (Thrombin). This should be done in concert with step 2 described above.
4. Source control
- By far, the most common cause of PPH is uterine atony (Tone). Therefore the first action taken should be physical maneuvers to improve tone. A bimanual uterine massage can be useful in stimulating uterine contractions. At the same time, one can evaluate for retained products of conception (Tissue). If tissue is felt, try to sweep out as much as possible while taking care to avoid uterine perforation. Note that this should be done with an empty bladder therefore a foley catheter should be placed prior to attempting massage.
- If the patient continues to bleed briskly, an effort can be made via balloon tamponade of the uterus, with a foley catheter (or, if available, a Bakri) with ~ 150 ml normal saline injected into the balloon. If the uterus is firm and bleeding continues it is reasonable to assess the genital tract for lacerations of the vaginal wall or cervix (Trauma). Cervical lacerations, ideally, should be repaired by an experienced obstetrician as this can have implications on future fertility. However, a vaginal laceration can be repaired just as a perineal laceration repair, taking care to approximate anatomy with absorbable sutures.
5. Administer uterotonics
- It is reasonable to begin uterotonic therapy in conjunction with uterine massage in a briskly bleeding patient. Oxytocin is first line and 10 U can be given immediately IM or as an infusion at 10 - 40 mU/minute to achieve and maintain uterine contractions. If hemorrhage is refractory to massage and oxytocin, continue pharmacotherapy for ongoing bleeding with Carboprost (Hemabate - 0.25 mg IM) and Misoprostol (cytotec – 1000 mcg per rectum).
Summary
Hemodynamically unstable PPH patients should be resuscitated like any other severely hemorrhaging patient. Utilizing a step wise approach as described above will help you maintain control of the situation. Notify an obstetrician. Resuscitate. Obtain adequate access. Source control. Administer uterotonics.
If the patient remains unstable, a myriad of other options exist with your interventional or obstetrical colleagues, including uterine artery embolization, ligation of uterine/internal iliac arteries, or hysterectomy.
Expert Commentary
This case outlines a common presentation of a postpartum hemorrhage in the ED.
A few points:
- While the patient in this scenario had just delivered, postpartum hemorrhage can occur days or even weeks following delivery/hospital discharge. Delayed postpartum hemorrhages are often caused by infection or retained products of conception, so if a patient is stable enough to perform a bedside scan, looking for retained products (which will show as echogenic on ultrasound) can be helpful. Ultimately, a patient with retained products is likely going to need to go to the OR for a D &C, so this is another reason to call OB/Gyn early.
- In the case of delayed postpartum hemorrhage, realize a patient may have been sent home following delivery with a fairly low hemoglobin, and may not have much reserve even given that pregnant and recently postpartum women have higher circulating blood volumes. She may have also lost a lot of blood prior to presentation, either that day or slowly over the past days/weeks.
- When interpreting DIC labs, remember that fibrinogen levels are higher in pregnant, as compared to nonpregnant, women. Thus, a ‘normal’ fibrinogen level may still represent a significant decrease. Most protocols for blood product resuscitation in the case of a postpartum hemorrhage involve replacement of clotting factors and fibrinogen along with packed red blood cells as DIC is fairly common with large volume blood losses.
- There are two goals of performing a bimanual exam: fundal massage and clot evacuation. Uterine atony will not improve if there is a large volume of clot in the uterus, so be aggressive about clearing these clots out.
- Uterine massage can be quite painful; if possible give either IV or IM narcotic (morphine, fentanyl, or dilaudid) prior to starting.
- Cervical lacerations and complex perineal lacerations often need to be repaired in the OR, either for better visualization or better pain control, often with either a spinal or epidural to help keep the patient comfortable and still. Packing the vagina with kerlix gauze while waiting for OB/Gyn can be one strategy to reduce bleeding.
- A good way to determine ongoing blood loss is to have someone weigh the chux with the blood on it (1 gram = 1 ml).
One other note: the box with risk factors for postpartum hemorrhage also include parity (the more
babies a woman has had, the higher the risk), a macrosomic fetus, and polyhydramnios.
Annie Dude, MD PhD
Maternal-Fetal Medicine Fellow, Northwestern Obstetrics & Gynecology
Posts You May Also Enjoy
How to cite this post
[Peer-Reviewed, Web Publication] Lang S, Macias M (2018, Feb 12). Post Partum Hemorrhage in the ED. [NUEM Blog. Expert Commentary By Dude A]. Retrieved from http://www.nuemblog.com/blog/post-partum-hemorrhage.
References
- MacDorman, M. F., Mathews, T. J., & Declercq, E. (n.d.). Trends in out-of-hospital births in the United States, 1990-2012.
- MacDorman, M. F., Mathews, T. J., & Declercq, E. R. (2012). Home births in the United States, 1990-2009. Hyattsville, MD: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics
- Marx, J. A., Hockberger, R. S., Walls, R. M., & Adams, J. (2002). Rosen's emergency medicine: Concepts and clinical practice. St. Louis: Mosby.
- Prevention and management of postpartum haemorrhage - RCOG. (n.d.). Retrieved July 23, 2016, from https://www.rcog.org.uk/globalassets/documents/guidelines/gt52postpartumhaemorrhage0411.pdf
- Anderson, J. M., MD, & Etches, D., MD. (n.d.). Prevention and Management of Postpartum Hemorrhage. Retrieved July 27, 2016, from http://www.aafp.org/afp/2007/0315/p875.html
Clinical Concept: Managing first trimester vaginal bleeding in the ED
Written by: Priyanka Sista, MD (NUEM PGY-2) Edited by: Spencer Lang, MD, (NUEM PGY-4) Expert
Commentary by: Natasha Wheaton, MD
Introduction
Vaginal bleeding in a pregnant patient occurs frequently (20-40% of all 1st-trimester pregnancies) and these patients present to the Emergency Department often. Patients in their 1st trimester (prior to 12 weeks gestational age) may not have initiated prenatal care and may not have a definitive intrauterine pregnancy (IUP). In fact, they may not be aware that they are pregnant at all. In patients with 1st trimester bleeding, consideration of ectopic pregnancy should be a priority for the emergency physician. Ectopic pregnancies, while rare, are responsible for the greatest morbidity and mortality in early pregnancy. When ectopic pregnancies are diagnosed - or even strongly suspected - management should involve consultation with an OB/GYN, either for emergent surgical management or medical management. The vast majority of 1st trimester bleeding, however, falls into a spectrum of “it’s too early to tell.” The differential of early bleeding with an undetermined pregnancy location ranges from completed abortions to benign subchorionic hemorrhages, with multiple options in between. This degree of uncertainty is often emotionally difficult to absorb for both patients and clinicians. Having a thorough management approach to 1st trimester bleeding, clearly communicating the uncertainty in prognosis, and providing accurate anticipatory guidance can better protect patients, ensure appropriate follow-up, and may even offer reassurance.
Diagnostic algorithm for vaginal bleeding in early pregnancy (< 12 weeks gestation age)
1. Is the patient hemodynamically stable?
If not, you should begin resuscitation and consult gynecology early for strong suspicion of ectopic pregnancy. Ensure the patient has 2 large-bore IVs, give crystalloids, check a hemoglobin, type and cross-match blood, and perform a FAST exam to look for free fluid in the abdomen. If the patient is actively hemorrhaging and you cannot wait for a type and cross, transfuse O-negative blood. Administer RhoGam (see below). If you suspect the patient is septic from a septic abortion, give antibiotics (see below). If you suspect the patient is headed to the OR emergently, order the appropriate pre-op labs.
Before continuing with the rest of the diagnostic algorithm, all pregnant vaginal bleeders will typically receive the following, unless there is rationale not to do so: urine pregnancy test, serum beta-hCG, type and screen, a CBC, and a transvaginal ultrasound.
2. Confirm an IUP with transvaginal ultrasound (TVUS).
For all intents and purposes, confirmation of an intrauterine pregnancy effectively rules out ectopic pregnancy (with rare exceptions, see heterotopic pregnancy, below).
How do you confirm an IUP?
With ultrasound:
Visualize a gestational sac (dark anechoic area) within the endometrial cavity. Typically visualized around 4-5 weeks gestational age.
Visualize a yolk sac (concentric circle, around 5.5 weeks) or fetal pole (6.5 weeks) within the gestational sac.
Measuring the Yolk Sac, a concentric circle within the gestational sac
Ectopic pregnancies secrete hormones that can cause changes in the endometrial lining, which may look like a gestational sac, called a “pseudogestational sac of ectopic pregnancy.” Therefore, an IUP is confirmed when the yolk sac or fetal pole is visualized within the gestational sac.
A and B: Endovaginal views showing an intrauterine pregnancy. The gestational sac (blue) is located within the endometrial stripe of the uterus (yellow) and contains a fetal pole (purple).
3. What if the TVUS is indeterminate and you cannot confirm an IUP?
If you cannot confirm an IUP by TVUS, then you cannot effectively rule out an ectopic pregnancy – this is deemed a pregnancy of unknown location (PUL). This means the patient may have an early presentation of ectopic pregnancy, may be too early in their pregnancy to visualize an IUP, or may have a nonviable IUP.
With an indeterminate TVUS, consider the serum beta-hCG level. The discriminatory zone is the level of beta-hCG above which one should be able to identify an IUP by ultrasound (for TVUS, the discriminatory zone 1000-1500 mIU/mL; for transabdominal ultrasound, it is 4000-6500 mIU/mL).
When patients have beta-hCG levels above the discriminatory zone, presume an ectopic pregnancy until proven otherwise and consult OB/GYN.
When patients have beta-hCG levels below the discriminatory zone and an indeterminate TVUS, you still cannot rule out ectopic pregnancy. Patients have presented with ruptured ectopic pregnancies with beta-hCG levels well below the discriminatory zone. If clinical suspicion is high or you identify free fluid in the pelvis, presume ectopic pregnancy and consult gynecology, regardless of beta-hCG level. If there is no free fluid identified and the patient remains stable and well-appearing, they will need close follow-up and monitoring in 48 hours to re-check serum-hCG levels. In normal viable pregnancies, the serum hCG is expected to roughly double every 48 hours in the 1st trimester. In ectopic or nonviable pregnancies, the serum hCG may seem normal, rise less quickly than expected, or in the case of spontaneous abortion, may plateau or fall. It is the ED physician’s responsibility to ensure this patient has good follow-up in 48 hours for serum beta-hCG recheck, as well as to give strict return precautions for signs of a ruptured ectopic pregnancy.
(A word on heterotopic pregnancy: Heterotropic pregnancy refers to pregnancies at two different implantation sites, most typically a combination of an IUP and an ectopic pregnancy. While it was and is quite rare, (1 per p 30,000 pregnancies), the incidence is increasing with the use of assisted reproduction techniques (ART). An estimated 1.5 per 1000 ART pregnancies are heterotopic. Therefore, patients who used ART should get a formal ultrasound in the ED, and all women should have close follow-up. )
4. Is the cervix open or closed? Is the pregnancy viable or nonviable?
When an IUP is confirmed, a patient with 1st trimester vaginal bleeding may be presenting with a spontaneous abortion. Classification of spontaneous abortion is provided in the table below, and requires a pelvic exam to determine whether the cervix is open or closed.
The majority of patients who present to the ED will be diagnosed with “threatened abortion,” which includes bleeding or cramping, a viable pregnancy, and a closed cervix. Approximately 50% of these patients will proceed to have a miscarriage.
Most spontaneous abortions in the 1st trimester will progress to completion without complications. If they do not progress, the patient may require medical or surgical intervention to remove all POC. Septic abortions may occur if infection complicates the pregnancy loss.
What makes a pregnancy nonviable? The following ultrasound findings:
Crown-rump length of > or = 7 mm and no heartbeat.
Mean sac diameter > or = 25 mm and no embryo
Absence of embryo w/ heartbeat > or = 2 weeks after a scan that showed a gestational sac without a yolk sac.
Absence of embryo with heartbeat > or = 11 days after a scan that showed a gestational sac with a yolk sac
Rh immunization
All Rh negative mothers should get 50 mcgs of RhoGam in the 1st trimester when they are less than 12 weeks gestation, based on expert clinical opinion. This will be good for any subsequent bleeding that occurs in the following 12 weeks. At some institutions, patients get the full dose RhoGam (300 mcgs) regardless of gestational age, but this is only recommended for patients beyond 12 weeks.
Counseling, FAQs, and Anticipatory Guidance
Given that implications of 1st trimester bleeding are often determined with time, it is critically important to establish appropriate follow-up plans for patients. Providing a safe, reassuring presence with clear information and clear instructions in the midst of uncertainty may help ensure appropriate follow-up. Here are some tips to keep in mind:
Ask all patients “Is this a desired pregnancy?” Do not make assumptions or judgments about the nuances of a patient’s situation.
Patients may mourn and grieve pregnancy loss similar to the loss of a child or family member. This can be true even for undesired pregnancies. We may forget that we need the same level of kindness for patients experiencing a spontaneous abortion as we do for the mother of a patient with fatal GSW injuries.
While we use the term “abortion” in a medical context, for most people it is fraught with political and emotional complexity. Please explain the medical term so that a patient does not see the word for the first time as a discharge diagnosis.
Pregnancy of Unknown Location Discharge Instructions:
1. Patients MUST follow-up in 48 hours to re-check a serum beta-hCG level.
What will happen at that visit? If there is an inappropriate rise in beta-hCG level, there is concern for ectopic or nonviable pregnancy. When the beta-hCG level is above the discriminatory zone, a repeat TVUS will be obtained to confirm a viable IUP. If there is no IUP determined, there is concern for ectopic or nonviable pregnancy. Gynecology will help in management decisions at that point: they may choose to monitor the hCG levels, or they may give you medications or consider operative management to help facilitate the nonviable pregnancy to its completion.
What will happen if you don’t follow-up? There is risk of the pregnancy growing in a dangerous location outside the uterus, rupturing, and causing life-threatening bleeding into the abdomen. There is also the risk of missed, incomplete, or septic abortions, which can be life-threatening if allowed to progress. Intervention is sometimes needed to help the body terminate an ectopic or nonviable pregnancy to avoid the possibility of life-threatening hemorrhage and/or infection.
What does a nonviable pregnancy mean? It means an abnormal pregnancy that will not be carried to completion due to some factors that make it incompatible with progression. (See above for diagnostic definitions)
2. Use Tylenol for abdominal cramping if needed. Stay well hydrated. Have 2 weeks of pelvic rest (nothing in the vagina for two weeks). There is no evidence to prove that pelvic rest improves outcomes or reduces the likelihood of spontaneous abortion. Theoretically, sexual activity may trigger ectopic rupture but there is no evidence to suggest this. However, pelvic rest is widely recommended to eliminate any possible feelings of guilt that patients may have regarding being responsible for a spontaneous abortion.
Patients with known open cervical os may continue to bleed significantly. Those with closed cervical os may develop an open os in the coming hours/days. Warn the patient that they may pass blood clots or products of conception. If a patient is concerned they passed products of conception, ask them to retrieve it and bring to their doctor’s or to the ED. Given them gloves and a specimen cup. If a patient is bleeding more than 2 pads an hour for more than 2 hours in a row, tell them to return to the ED.
3. You must immediately return to the emergency department if you have:
Increased or persistent abdominal pain
Heavy vaginal bleeding (more than 2 pads an hour for 2 consecutive hours)
Weakness, dizziness, lightheadedness.
FAQs, and answers to have in your back pocket:
1. How common is 1st trimester bleeding? 25-33% of pregnancies will have 1st trimester bleeding.
2. What does this mean regarding the future of this pregnancy? Approximately half of patients with 1st trimester bleeding will have a subsequent spontaneous miscarriage.
3. Does this mean I’m miscarrying? Unless the patient has an open os or a documented nonviable pregnancy, it is difficult to tell in the ED. It’s okay to say “I don’t yet know,” and “time will tell, which is why follow-up is important.”
4. What do I need to watch out for? What activities can I resume or not resume? With any vaginal bleeding in pregnancy, suggest pelvic rest for 2 weeks. Avoid ibuprofen; take Tylenol for pain. Watch out for the ectopic precautions listed above. Otherwise, if you feel up for doing something, it is OK to do it.
5. How much bleeding is too much? More than 2 pads an hour for more than 2 hours is too much bleeding.
6. Why did this happen? Miscarriages are common, occurring in 25-33% of pregnancies. More than half the time, miscarriages occur due to an intrinsic genetic or chromosomal abnormality that occurs during fertilization, rather than due to some inheritable trait, or extrinsic factor. For patients who have recurrent miscarriages, there may be structural or extrinsic causes for pregnancy termination, which warrants workup by the gynecologist.
7. Did I miscarry because I had sex / took Tylenol / went on a run / had a glass of wine before I realized I was pregnant? Reassure the patients that miscarriage is common and that there’s nothing they did to cause this. Miscarriage happens in up to one-third of all pregnancies.
8. Who gets genetic testing and has tissue pathology testing? For a first-time miscarriage, no genetic testing or tissue pathology is warranted. For subsequent miscarriages, products of conception can be sent to tissue pathology, and extrinsic causes of spontaneous abortion can be evaluated by a gynecologist.
9. What does a miscarriage mean for future pregnancies? Most patients will subsequently have normal future pregnancies., but it is true that having a miscarriage increases the chance of subsequent miscarriages slightly.
10. When can I start trying to get pregnant again? Patients can ovulate and become pregnant as soon as 2 weeks after miscarriage. Most gynecologists recommend waiting one menstrual cycle so that it is easier to calculate the due date of the next pregnancy, but there is no medical reason to wait to begin trying again. If patients do not want to get pregnant again right away, be sure to use birth control.
11. Reassure patients that if they are concerned about anything at all, or there is any change in their health, there should be a low threshold for calling a gynecologist on-call or returning to the ED for re-evaluation.
Expert Commentary
This is a comprehensive and well written entry on a very common complaint seen in the ED. Though the basic principles of early pregnancy related vaginal bleeding are relatively straightforward; confirm an IUP, administer Rhogam if indicated and provide anticipatory guidance, there are several nuances that bear mentioning and will improve patient care as well as patient experience.
As stated in the blog, after determining hemodynamic stability, the next step is usually to ensure the pregnancy is developing in the correct anatomic location (ie “find the IUP!”). However, there are some nuances regarding transvaginal ultrasound in early pregnancy. First, it is worth emphasizing that ectopics can exist at truly ANY hcg level (fine, fine except at zero) so don’t let anyone convince you to skip the ultrasound because “the beta is too low”. In fact, in a retrospective study of 730 women seen in the ED with early pregnancy bleeding, a β-HCG of <1,500 mIU/mL more than doubled their chances of having an ectopic pregnancy (Acad. Emerg. Med. 2003;10:119-26). Another study found that 40% of ruptured ectopics had β-HCG < 1,000 mIU/mL, suggesting that those women with ectopics and lower quants may actually have poorer outcomes.
If you are doing your own transvaginal ultrasound (or even when reviewing those done by radiology), don’t confuse a pseudogestational sac for a true gestational sac. Pseudosacs are seen in up to 20% of ectopic pregnancies (Radiology. 1979;133:451–454) and are irregular, heterogeneous and lack a yolk sac. When you are concerned for a pseudosac, be especially vigilant for secondary signs of ectopic including free fluid in Douglas’ pouch posterior to the uterus or abnormal adnexal masses. Finally, remember that while the vast majority of ectopics are tubal, there are multiple other locations where ectopics can implant some of which are particularly difficult to detect on ultrasound. Interstitial ectopics are particularly difficult as they appear to be in the uterus but are in fact within the myometrial tissue rather than in the uterine cavity. Though the exact measurements remain controversial, most agree that a measurement less than 8 mm of myometrium surrounding the gestational sac is abnormal (https://radiopaedia.org/articles/interstitial-ectopic-pregnancy).
Though there is some data suggesting that pelvic exams in the ED often don’t alter management, I still advocate for a pelvic exam in these patients for several reasons. First, it helps quantify the amount of bleeding. There is some data that those with heavy bleeding are more likely to miscarry than those with lighter bleeding (Hasan et al, Obstet Gynecol. 2005 Nov;106(5 Pt 1):993-9). If there are products present at the cervical os, delivery of those with ring forceps may shorten the patients pain and bleeding. An evaluation of the cervix may rule out other causes of bleeding including STIs or trauma. Remember that domestic violence levels peak during pregnancy so be on the lookout for it, GU trauma can be a sign! Finally, a bimanual exam can help risk stratify the patient and aid in your counseling. If the cervix is dilated, that is a nonviable pregnancy and an “inevitable abortion”. The counseling for that woman is significantly different than counseling for one with mild vaginal spotting and a closed cervical os.
A few notes on the practicalities of performing this physical exam. First, a pelvic can be particularly uncomfortable when pregnant so please take your time and warn the woman before you start. Be ready to collect tissue so you’re not scrambling if you find tissue unexpectedly. Grab ring forceps, a sterile collection cup as well as Fox swabs and an extra Chux to cover your used instruments. If you collect tissue, it may be worth sending to the lab for chromosomal analysis though this is really only indicated in recurrent pregnancy loss.
A word on ART patients. First, know that their dates are exact so if you don’t see a yolk sac when expected (5.5 weeks) or a heartbeat when expected (just under 6 weeks) it’s probably not a viable pregnancy. Also, be highly suspicious of hetero-ectopics in these patients! Their risk is upwards of 2-5% (Fertil Res Pract. 2015; 1: 15.) so don’t be too comforted by a TVUS showing an IUP. I would advocate for early involvement of their REI physicians to ensure close and appropriate early follow up.
As we wrap up, let’s talk numbers. While there is some data showing a 50% miscarriage rate for all-comers to the ED with vaginal bleeding in early pregnancy (< 12 weeks), this number changes significantly depending on what we find in the ED. With a closed cervix, mild bleeding and a TVUS showing a heartbeat, the risk drops to closer to 10%. The heavier the bleeding, the higher the risk. Finally, a subchorionic hemorrhage on ultrasound does increase the risk of miscarriage to closer to 25% though the risk also depends on the size of the hemorrhage (J Obstet Gynaecol. 2006 Nov;26(8):782-4.)
So, what about follow up? As the blog notes, if we have yet to confirm an IUP, these patients need 48-hour follow up for a repeat β-HCG and consideration of an ultrasound. But what if we did find an IUP? If the woman does go on to miscarry, does she need to see a provider? And how long can she bleed before Ob/Gyn would consider offering her surgical intervention (ie D&C) if desired by the patient? Does she have to have a D&C?
In general, I counsel women with documented IUPs that if they begin to bleed significantly more, pass clots or tissue and go on to have more intense cramping they are likely to be experiencing a miscarriage. They do not necessarily need to return to care as many women prefer to miscarry in the comfort of their own homes, as long as there is not excessive bleeding, severe abdominal pain, fainting or other severe symptoms. I warn her (and her partner if present) that the cramping will be significant and she will pass clots and tissue but the pain should not be persistent and/or severe. Finally, I counsel women that she will likely have bleeding for 1-2 weeks but it should slow after several days. If it does not and she continues to have pain and/or heavy bleeding at the 1 week mark she should follow up with a provider for a repeat ultrasound to ensure she does not have retained products of conception and discussion of possible D&C versus continued expectant management.
Lastly, some comments on the words we use in these cases; it matters! Though this is “bread and butter” emergency medicine for us, this day will likely stick in this patient’s, and her families’, minds for the rest of their lives. Do all those things we were taught in medical school when breaking bad news; Sit down, take your time and make eye contact. Say you’re sorry this is happening and you’re sorry there are often no black or white answers in these cases. Don’t use the word abortion and if those words are in her discharge instructions explain why. At the end of my counseling I always tell the woman that it’s not her fault and that there was nothing she could have done to prevent this or prevent an ultimate miscarriage if it does occur. I say “Pregnancy is a very complicated process and it doesn’t always go right. Usually pregnancies that miscarry this early because the baby wasn’t developing normally and would have never developed into a healthy baby”. Be the doctor you would want for your family member in this sensitive, scary and potentially life-altering time!
Natasha Wheaton, MD
Associate Program Director, UCLA Emergency Medicine
Posts You May Also Enjoy
How to cite this post
[Peer-Reviewed, Web Publication] Sista P, Lang S (2017, Dec 18). Clinical Concept: Managing first trimester vaginal bleeding in the ED. [NUEM Blog. Expert Review By Wheaton N]. Retrieved from http://www.nuemblog.com/blog/1st-tri-bleed
Ovarian Hyperstimulation: Not Your Ovarian Average Cyst
Pregnancies resulting from assisted reproduction are more complicated, with higher rates of ectopic, heterotopic and multifetal pregnancies, in addition to higher rates of abortions and premature deliveries. Other complications include venous thromboembolism as well as ovarian hyperstimulation syndrome. These are high risk pregnancies that will present to your emergency department and you should be aware of key management principles.