Written by: Andy Rogers, MD (NUEM PGY-2) Edited by: Dana Loke, MD (NUEM PGY-4) Expert commentary by: Quentin Reuter, MD
Seeing double: toil and trouble?
Introduction
Double vision, or diplopia, is a relatively infrequent presenting symptom in the emergency setting, representing 0.1% of Emergency Department (ED) complaints (1). Diplopia can result from benign processes, such as dry eyes or idiopathic cranial nerve palsy, to emergent conditions with high morbidity, such as stroke, aneurysm, or inflammatory processes. Given a wide range of possible outcomes for a less common presenting complaint, it is worth reviewing the neuroanatomy and etiologies of diplopia, as well as a generalized approach to the patient presenting to the ED with double vision.
Neuroanatomy
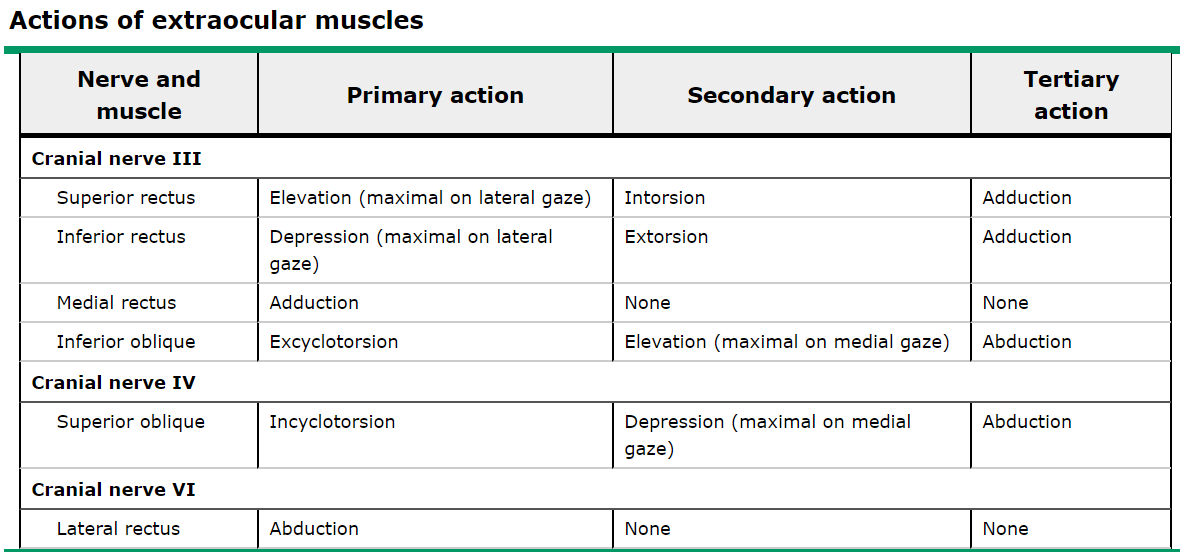
The neuroanatomy underlying control of the extraocular muscles and their ability to provide gaze alignment is complex and worth a brief review. Eye movements are governed by six extraocular muscles, which are controlled by four cranial nerves, summarized in Table 1 and Figure 1 below. The nuclei for the cranial nerves are located in the brainstem. The nerves course from the brainstem, through the subarachnoid space, the cavernous sinus (Figure 2), the orbital apex, and finally to their respective extraocular muscles. Given the close proximity of nearby structures, lesions can often be localized based on associated symptoms, in addition to gaze palsies, to help guide workup and diagnosis. For instance, an important additional function of CN III includes the parasympathetic fibers that travel along the oculomotor nerve that contribute to pupillary constriction.
Table 1: Summary of functions of the extraocular muscles, grouped by cranial nerve. (2)
Figure 1: Diagnostic positions of gaze with associated extraocular muscles contributing to movement. (3)
Figure 2: Cavernous sinus and its contents. Note that the cavernous sinus is symmetric about the pituitary fossa (only one side is shown above) (4)
Initial approach to diplopia
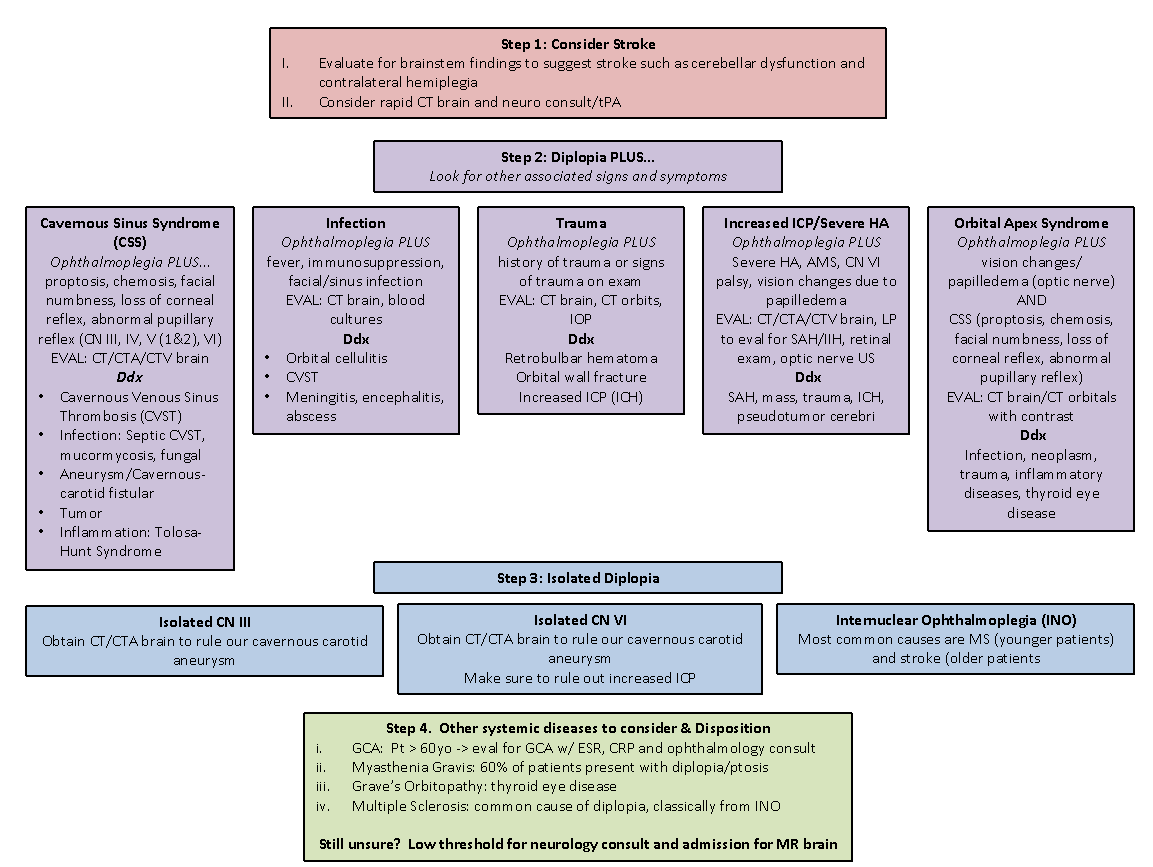
Dr. Margolin and Dr. Lam published an excellent review of the approach to diplopia in the ED, summarized in Figure 3 (5). Critical to the diagnosis is a good history and neurologic exam. Their approach involves the follow steps:
Determine if symptoms are monocular or binocular
Determine if there are associated neurologic signs or symptoms
If isolated diplopia, determine if the palsy localizes to a third or sixth nerve palsy, or if it is a complex motility disorder
Screen for giant cell arteritis (also known as temporal arteritis) in all patients over the age of 60
Figure 3: Approach to patient with diplopia, from Margolin and Lam (5)
Monocular or Binocular
The first step in the approach to diplopia in the ED is to determine if the diplopia is monocular or binocular. Some patients may not know or not have checked prior to presentation. Ask the patient “does the double vision resolve when you close one eye?”
Monocular Diplopia
Monocular diplopia persists with one eye closed. This localizes the lesions to the affected eye and reflects that the issue is not with misalignment of gaze. It is almost always benign and most often due to dry eyes and refractive error (5, 6). Referral to an ophthalmologist is appropriate and no further imaging is indicated unless warranted by other features of the patient’s presentation.
Binocular diplopia
Binocular diplopia resolves with one eye closed. This indicates ocular misalignment that can be due to an issue with the muscle, nerve, or CNS.
Binocular diplopia with associated neurologic signs
Diplopia with other associated neurologic signs is concerning. Acute onset of symptoms may be due to intracranial hemorrhage, cerebrovascular disease, or rapidly progressive neurologic disorder. Initiation of stroke protocol is valuable for several reasons. It allows for rapid diagnosis of hemorrhagic or ischemic stroke, evaluates for cerebral aneurysm, and involves Neurology quickly in the patient’s care. Some neurologic signs and symptoms, or clusters of signs and symptoms can help identify the etiology (5-7). See Table 2 below.
Binocular diplopia without associated neurologic signs (Isolated diplopia)
In all patients presenting with diplopia, careful examination of the extraocular movements and pupils are important to localizing the lesion. Note that diplopia can be due to weakness in one direction or entrapment of the muscle limiting range of motion. Both etiologies must be kept in mind. Figure 1 is a good reference to identify what muscles and nerves may be affected based on directional limitations in extraocular movements. Some important questions to ask include (6):
Are the two images side by side, on top of each other, or on a diagonal? – helps to tease out the plane of action of the affected muscles and their respective nerves
What field of gaze makes the double vision worse? – this represents the field of vision of a paretic muscle or opposite the field of action of a restricted muscle
Can you move your head to correct the vision? – Oblique diplopia due to CN IV paresis often can be distinguished with tilting of the head. Vertical diplopia can be improved with neck extension or flexion.
Is there pain with eye movement? – suggests myopathy or orbital process
Isolated 4th nerve palsies:
The trochlear nerve innervates the superior oblique muscle. Patients often complain of vertical or diagonal diplopia that may correct with head tilt. CN IV palsies are most commonly due to trauma or are idiopathic in nature (6). In idiopathic CN IV palsies, the patient should be referred to ophthalmology. The palsy often resolves within two weeks. Important neurologic signs to look for with a CN IV palsy are cerebellar signs. The trochlear nerve exits on the dorsum of the brainstem and may be compressed by a posterior fossa tumor.
Isolated 6th nerve palsies:
The abducens nerve, or 6th cranial nerve, innervates the lateral rectus muscle. In a 6th nerve plasy, patients will complain of a horizontal diplopia. There is often inward deviation of the affected eye (esotropia) and symptoms are made worse with lateral gaze to the affected side (6). This nerve palsy is often idiopathic in etiology, with diabetes mellitus as a risk factor. Be careful to assess for bilateral 6th nerve palsy. The abducens nerve has a long, isolated course intracranially; tumors can affect the bilateral abducens nerve before other areas of the brain are affected.
Isolated 3rd nerve palsy:
The oculomotor nerve innervates four muscles and carries parasympathetic fibers that control pupil constriction. Its course is closely related to the posterior cerebral artery and posterior communicating artery. A palsy of the 3rd nerve (especially with pupillary involvement) may be due to cerebral aneurysm and must emergently be evaluated with CTA, MRA, or intravascular angiography. An acute isolated 3rd nerve palsy may be due to expanding aneurysm that is at risk of imminent rupture (7). Pupil sparing 3rd nerve is rarely due to an aneurysm and more often ischemic injury (6,7).
Internuclear ophthalmoplegia (INO)
In the setting of horizonal diplopia, also look for internuclear ophthalmoplegia (INO). An INO is impaired horizontal movement with weak adduction of the affected eye and abduction nystagmus of contralateral eye (8). This localizes the lesion to the medial longitudinal fasciculous (MLF) in the dorsomedial brainstem tegmentum. The MLF connects the 6th nerve nucleus and medial rectus subnucleus of the 3rd nerve nucleus to coordinate lateral conjugate gaze movement. In patients <45 years old, it is most commonly caused by multiple sclerosis and is often bilateral (73%) (9). In older patients, especially those with vascular risk factors, it is caused by cerebrovascular disease and is usually unilateral. Up to a third have other causes, including infection, tumor, trauma, myasthenia gravis, and Guillain-Barre. Look for historical clues and other neurologic exam findings. Presence of an INO requires MRI workup.
Complex motility abnormality
If the diplopia doesn’t isolate to a specific cranial nerve, consider what nerves may be involved and where they are close together to consider anatomic abnormalities. This includes the cavernous sinus, orbital apex, and brainstem. Brainstem lesions will often have other neurologic deficits identified on exam.
Cavernous sinus
Cavernous sinus lesions will affect multiple cranial nerves but should not affect visual acuity as the optic nerve does not pass through in relation to the other structures (5,7). The pituitary gland also resides between the cavernous sinuses. Pituitary mass or apoplexy can compress laterally causing ophthalmoplegia. Another key concern is septic cavernous sinus thrombosis. These patients will often be septic and febrile. Conversely, consider asking your septic patients if they have double vision! Consider CT and CT venogram of brain and orbits.
Orbital apex
The orbital apex involves all extraocular muscles, sympathetic fibers, and cranial nerves 2/3/4/6/V1/V2. Here, the optic nerve is in close anatomic relation to the nerves and muscles of ocular motility. Any ophthalmoplegia with decreased vision or numbness in V1 or V2 distribution should raise concern for orbital apex pathology (5,7). Consider CT with contrast of the orbits.
Giant Cell Arteritis
Giant cell arteritis (also known as temporal arteritis) is an important diagnosis to always consider in an elderly patient presenting with diplopia. Missing this diagnosis can lead to permanent vision loss. Diplopia is actually an uncommon finding in GCA, occurring in roughly 5% of cases. However, diplopia has the second highest positive likelihood ratio (LR 3.4) for GCA (the highest being jaw claudication – LR 4.2) (10). In an elderly patient presenting with diplopia, be on the lookout for other signs and symptoms suggestive of GCA, such as jaw claudication, fevers, vision loss, temporal headaches, PMR, or elevated inflammatory markers.
Key Points
Diplopia is a relatively rare presenting complaint in the Emergency Department, and it can portend a wide range of disease, from the benign to the emergent
A good history and physical exam are key to diagnosis
Use your physical exam and the presence of other neurologic signs and symptoms to try to localize the lesion and guide imaging choice
Screen for temporal arteritis in the >60 population
Expert Commentary
Diplopia is a rare but potentially dangerous chief complaint, making up approximately 0.1% of all ED visits. [1] In one study of 260 ED patients with non-traumatic, binocular diplopia, 64% had primary diplopia (i.e. no identifiable cause found, likely from microvascular ischemic disease) and 36% had secondary diplopia (i.e. caused by some discernible pathology). Of patients with secondary diplopia, stroke accounted for nearly 50%, with multiple sclerosis (MS), tumor, aneurysm, myasthenia gravis (MG), and carotid cavernous fistula (CCF) accounting for the other diagnoses. [1] Given the dangerous etiologies at play, clinicians must approach these patients in a systematic and cautious manner. (Figure 1)
In patients presenting with diplopia, the first concern must be the possibility of stroke and the need to consider time-sensitive treatment with thrombolytics. Cranial nerves (CN) 3, 4, and 6 are supplied by the vertebrobasilar arterial system and strokes affecting these nerves most often present with cerebellar dysfunction and/or “crossed signs” or contralateral hemiparesis from involvement of the corticospinal tracts. [2] Thrombolytic treatment must be considered if the patient is presenting within the appropriate time window and stroke is suspected. If the patient is outside of the tPA window but suspicion remains for an ischemic process, these patients may benefit from neurologic consultation and MR imaging.
After considering stroke, look for other associated signs or symptoms that may lead to the correct diagnosis. If a patient has fevers, facial infections, or meningismus one must consider orbital cellulitis, cavernous venous sinus thrombosis (CVST), meningitis, or encephalitis. If a patient had a traumatic injury, consider orbital wall fractures, retrobulbar hematoma, increased intracranial pressure (ICP), and intracranial hemorrhage. If a patient has proptosis, chemosis, headaches, and/or facial sensory changes, consider cavernous sinus pathology such as CCF, aneurysm, CVST, mass, or Tolosa-Hunt syndrome (idiopathic inflammatory changes within the cavernous sinus). If a patient has a severe headache, CN 6 palsy, and/or AMS, consider subarachnoid hemorrhage or other causes of increased ICP. Notably, up to 5% of ruptured PCOM aneurysms present with a CN 6 palsy. [3] Lastly, if patients have concomitant vision changes/loss, consider pathology of the orbital apex such as mass, infection, and thyroid eye disease.
If no associated signs or symptoms exists, evaluate the patient for an isolated CN palsy. (Figure 2) The Margolin paper discusses the importance of obtaining a CTA brain in patients with isolated CN 3 palsy to rule out an intracranial aneurysm. I would also highlight the importance of obtaining a CTA when patients have an isolated CN 6 palsy as 16% of patients with cavernous sinus carotid aneurysm presented with isolated CN 6 palsy in one study. [4] In patients with internuclear ophthalmoplegia (INO), admission for MR and neurology consultation is appropriate to rule out stroke and MS, the most common causative pathologies. Finally, clinicians must consider systemic disease entities such as giant cell arteritis (GCA), MS, and MG. GCA is a vision-threatening disease and must be considered in patients over the age of 60 with transient or persistent diplopia as it is a presenting complaint in 6-27% GCA patients. [5]
Finally, a word of caution when approaching patients with diplopia. The Margolin article suggests that some patients with isolated diplopia can have non-emergent outpatient follow-up once an aneurysm has been excluded. Others have also recommended that a CT brain is not useful in the setting of isolated diplopia citing one study showing CT had a sensitivity of 0% in 11 patients with isolated secondary diplopia as the rationale. [6] I believe these recommendations are unrealistic for many reasons. First, diplopia is encountered only infrequently in the ED, making the gains of avoiding a CT brain and inpatient neurologic workup minimal. Furthermore, the neuro-ophthalmologic exam is challenging, and some patients can present with partial palsies or deficits involving multiple cranial nerves, making diagnosis of a specific CN challenging. Clinicians must also be 100% confident that no other signs or symptoms exist such as jaw claudication in GCA or subtle vision changes and papilledema for pseudotumor cerebri prior to discharge, a difficult task in a busy ED. Ensuring reliable and urgent neurologic follow-up and outpatient MR imaging can also be difficult or impossible in many health systems. Patients will also likely appreciate obtaining a definitive diagnosis for a concerning neurologic symptom like diplopia in a timely manner. Moreover, of the 11 patients with isolated secondary diplopia for which CT was not useful in the Nazerian article, two had strokes, another patient had a mass, and another had a CFF that the CT missed. [1] Diagnoses such as stroke, aneurysm, MS, GCA, and neoplasm can all have morbid ramifications if not expediently diagnosed.
As such, in patients with isolated diplopia, it may be appropriate for ED clinicians to rule out aneurysm with vascular imaging and admit for neurology consultation and MR to evaluate for more sinister pathology. ED clinicians should at the very least discuss the case with a consulting neurologist to ensure appropriate management. While the majority of patients with isolated diplopia will go on to be diagnosed with microangiopathic ischemia, given the above concerns, it would seem reasonable for ED clinicians to err on the side of caution and get help from their local neurology colleagues for these challenging patients.
Citations:
1. Nazerian P, Vanni S, Tarocchi C, et al. Causes of diplopia in the emergency department: diagnostic accuracy of clinical assessment and of head computed tomography. Eur J Emerg Med 2014;21:118-24.
2. Rowe F, UK VISg. Prevalence of ocular motor cranial nerve palsy and associations following stroke. Eye (Lond) 2011;25:881-7.
3. Burkhardt JK, Winkler EA, Lasker GF, Yue JK, Lawton MT. Isolated abducens nerve palsy associated with subarachnoid hemorrhage: a localizing sign of ruptured posterior inferior cerebellar artery aneurysms. J Neurosurg 2018;128:1830-8.
4. Stiebel-Kalish H, Kalish Y, Bar-On RH, et al. Presentation, natural history, and management of carotid cavernous aneurysms. Neurosurgery 2005;57:850-7; discussion -7.
5. Haering M, Holbro A, Todorova MG, et al. Incidence and prognostic implications of diplopia in patients with giant cell arteritis. J Rheumatol 2014;41:1562-4.
6.Kisilevsky E, Kaplan A, Micieli J, McGowan M, Mackinnon D, Margolin E. Computed tomography only useful for selected patients presenting with primary eye complaints in the emergency department. Am J Emerg Med 2018;36:162-4.
Figure 1: Approach to the patient with diplopia
Figure 2: Clinical exam for CN 3 palsy, CN 6 palsy, and INO
Figure 2A: Complete CN 3 palsy
Figure 2B: Complete CN 6 palsy
Figure 2C: Internuclear Ophthalmoplegia
Quentin Reuter, MD
Assistant Professor of Emergency Medicine
Northeast Ohio Medical University
Citations
Sources:
“Causes of Diplopia in the Emergency Department: Diagnostic Accuracy of Clinical Assessment and of Head Computed Tomography.” Nazerian et al. European Journal of Emergency Medicine. 21 April 2014 (2):118-24. Doi 10.097/MEJ.0b01323283636120
“Actions of Extraocular Muscles.” UpToDate.com. Accessed 11/22/18.
Image adapted from: “Diagnostic Positions of Gaze.” UpToDate.com Accessed 11/22/18.
Image from: “ZSFG Neuro Report: Multiple Cranial Neuropathies – Spotlight on the Cavernous Sinus.” Stern, Rachel. UCSF Internal Medicine Chief Resident Hub. Published 28 Oct 2016. Accessed 20 November 2018. https://ucsfmed.wordpress.com/2016/10/28/zsfg-neuro-report-multiple-cranial-neuropathies-spotlight-on-the-cavernous-sinus/
“Approach to a Patient with Diplopia in the Emergency Department.” Margolin, Edward and Lam, Cindy. Journal of Emergency Medicine. Volume 54, Issue 6. June 2018. pp799-806. Accessed 16 November 2018.
“Overview of Diplopia.” Bienfang, Don. UpToDate. Last updated 20 June 2017. Accessed 17 November 2018.
“Third Cranial Nerve (Oculomotor Nerve) Palsy in Adults.” Lee, Andrew. UpToDate. Last updated 19 June 2017. Accessed 17 November 2018.
“Internuclear Ophthalmoparesis.” Frohman, Teresa; Frohman, Elliot. UpToDate. Last updated 4 December 2017. Accessed 18 November 2018.
“Internuclear Ophthalmoplegia Unusual Causes in 114 of 410 Patients.” Keane, James. Arch Neurol. 2005;62(5):714–717. doi:10.1001/archneur.62.5.714
“Does This Patient Have Temporal Arteritis?” Smetana, Gerald; Shmerling, Robert. JAMA. 2002;287(1):92–101. doi:10.1001/jama.287.1.92
How To Cite This Post
[Peer-Reviewed, Web Publication] Rogers A, Loke D. (2019, Nov 18). Approach to Double Vision in the ED. [NUEM Blog. Expert Commentary by Reuter Q]. Retrieved from http://www.nuemblog.com/blog/double-vision.