Written by: Philip Jackson, MD (NUEM ‘20) Edited by: Logan Weygandt, MD, MPH NUEM ‘17) Expert Commentary by: Katie Colton, MD
Relying on in-house radiology reads of imaging is a habit that EM trainees are encouraged to avoid, but one that can be appealing when practicing in a busy, large academic facility with 24-hour radiologist staffing. By reading one’s own images, not only do EM physicians gain skills in diagnostic radiology, which they can employ when an attending radiology read is not readily available but more importantly, the EM physician can correlate history and physical with imaging and help detect subtle pathology. Recent studies have shown that even attending EM physicians are often deficient in reading non-contrast CT scans of the head, however, with minimal training residents have been shown to make significant improvements. [2,3]
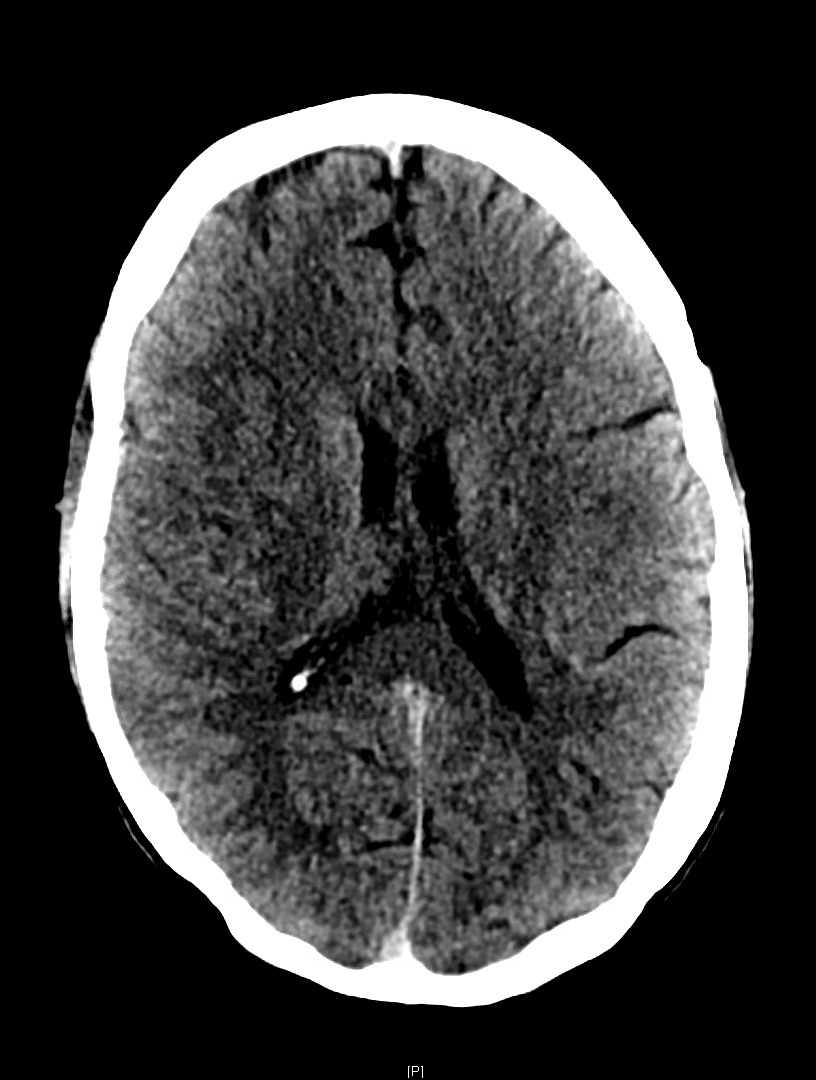
An elderly male with a history of hypertension and Fuch’s corneal dystrophy presented to our ED the morning after developing acute on chronic worsening of the blurry vision in his R eye. He suffered from persistent blurry vision but stated that it had suddenly worsened while watching TV the previous night. He then developed a left-sided occipital headache that continued through the following morning. He also noticed that his thinking was “cloudy” and despite being a healthcare professional could not describe his own medical history or list of medications. He described blurriness especially on the right. On visual field confrontation, the patient was found to have a binocular R sided superior quadrantanopsia. The rest of his neurologic exam was unremarkable. As these findings were concerning for stroke specifically in the left temporooccipital region known as Myer’s loop, we obtained a STAT non-contrast head CT.
As the so-called green arrow-signs on the CT image indicate, there was indeed a significant amount of cerebral edema present in the L temporal lobe white matter, which contains the anterior optic radiations carrying information from the R superior visual field and corresponds to our patient’s deficit. Upon discovering this lesion, our team immediately called our radiology colleagues who confirmed our concern for an acute ischemic infarct.
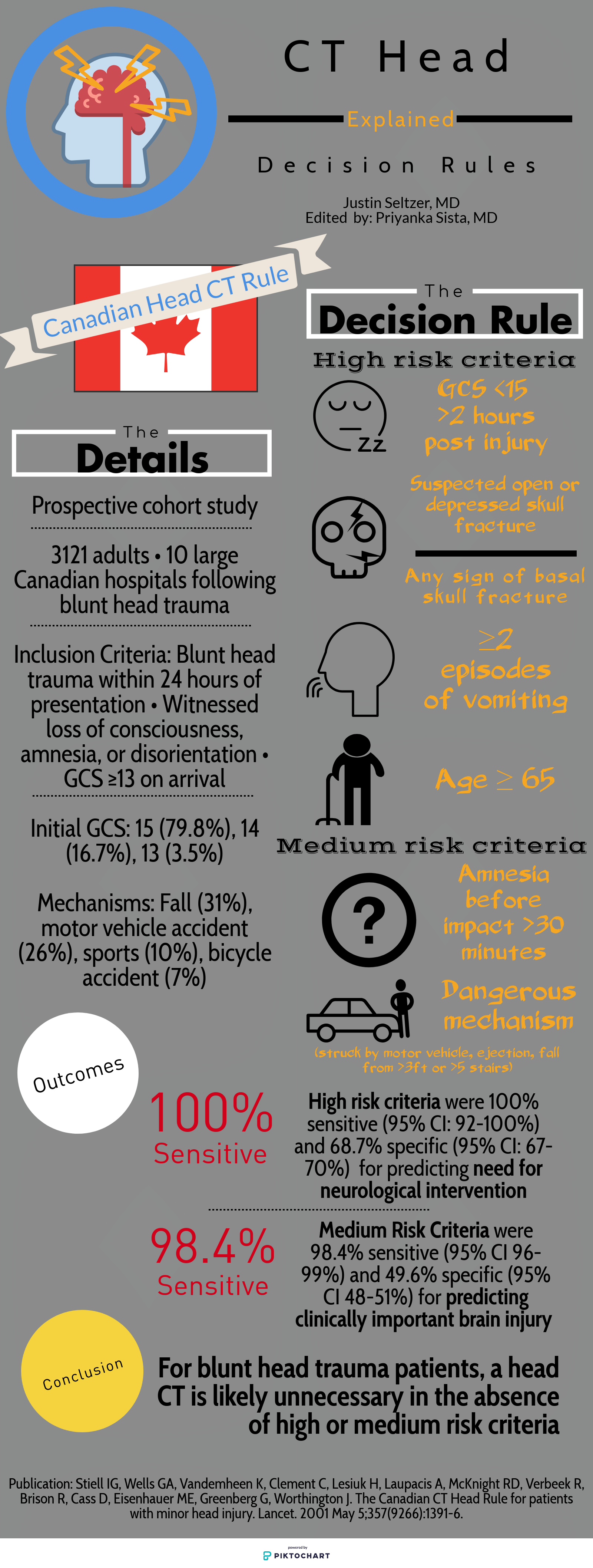
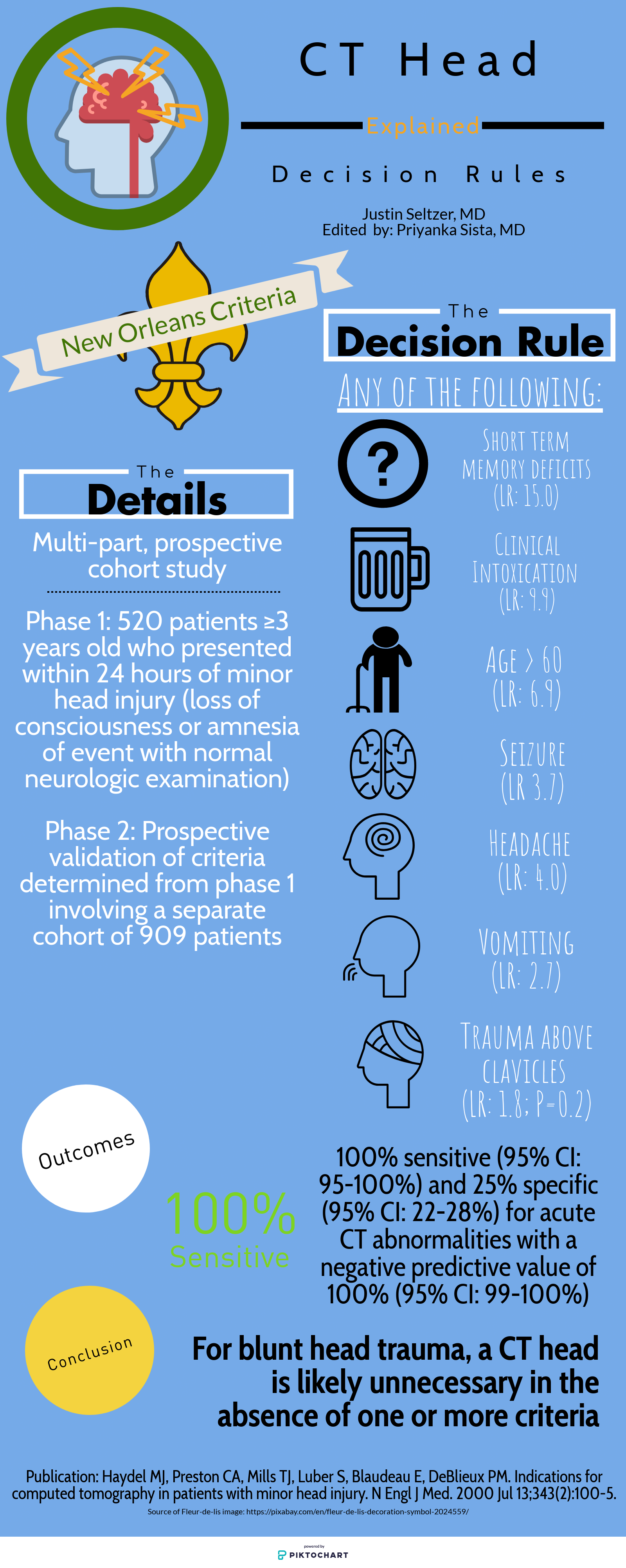
Like any other task in the ED, reading a head CT should be conducted as efficiently and accurately as possible using a standardized approach. EM residents have been found to be somewhat deficient in our ability to evaluate noncontrast head CTs; however, studies have shown that with adequate training, our skills can significantly improve. [3] Perron et al describe the simple but systematic approach “Blood Can Be Very Bad.” This mnemonic reminds residents to examine for the presence Blood, the shape and consistency of the Cisterns, the texture of the Brain parenchyma, the Ventricles, and the presence of fractures and symmetry of the Bony structures.
Blood: In a non-contrast CT, blood will appear as hyperdense (bright/white) fluid. As blood ages over weeks, it will become increasingly hypodense (darker). Blood will present in one of the four following ways:
Subarachnoid hemorrhage - A dreaded complication of trauma, a ruptured aneurysm, or an arteriovenous malformation can lead to blood pooling in gravity-dependent areas correlating with the particular arterial defect. Rupture of the anterior communicating artery (ACA) will distribute blood in and around the interhemispheric fissure, suprasellar cistern, and brainstem. Rupture of the middle cerebral artery (MCA) will distribute blood in the Sylvian and suprasellar cistern, while the posterior cerebral artery (PCA) will also distribute in the suprasellar cistern.
Subdural hemorrhage (SDH) – Caused by rupture of the bridging veins, SDHs will present as a crescentic lesions that often cross suture lines. SDHs can be acute, chronic, or mixed, and thus will have varying degrees of density.
Epidural Hemorrhage - Another serious complication of trauma, epidural hemorrhages will present as a lenticular (biconvex) areas of hyper-attenuation. Caused by arterial laceration, with the most common being the middle meningeal artery, epidural hemorrhages can rapidly expand and cause significant and rapid mass effect. Early identification is thus crucial to reducing mortality from these injuries.
Intraparenchymal/intraventricular hemorrhage - Often the result of hypertensive disease in elderly patients or as hemorrhagic strokes, intraparenchymal hemorrhage will most often be located in the basal ganglia. Amyloid angiopathy (associated with Alzheimer’s dementia) often presents as wedge-shaped areas of hemorrhage in the outer cortex. Trauma leading to brain contusion can also present with intraparenchymal hemorrhage. All intraparenchymal hemorrhages (as well as subarachnoid hemorrhages) can potentially rupture into ventricles causing intraventricular hemorrhage and resultant hydrocephalus.
Cisterns: Cisterns are spaces surrounding and cushioning brain matter with cerebrospinal fluid. Each of the four major cisterns should be examined for blood or signs of mass effect: the sylvan fissure (in between temporal and parietal lobes), the circummesencephalic or peripontine cistern, the suprasellar (surrounding the circle of Willis), and the quadrigeminal (atop the midbrain).
Brain matter: Always examine the gyri for and for distinct grey-white matter differentiation. Ischemic strokes, as in our case, will present with blurring of the grey-white differentiation and cerebral edema (areas of hypodensity). Early strokes may not be apparent on CT, but after 6 or more hours hypodense lesions should be present with maximal edema occurring approximately 3-5 days after the event. Always examine the falx for midline shift through multiple slices.
Ventricles: Examining the third and fourth ventricles is crucial in determining the presence of blood hydrocephalus (dilation) or mass effect (asymmetry).
Bone: The bony structures of the head should all be examined for fractures, especially depressed skull fractures, which usually denote intracranial pathology. Also, examining the sphenoid, maxillary, ethmoid, and frontal sinuses for air fluid levels should raise suspicion for a skull fracture. Separate bony windows are available for close examination of these high-density structures. [1]
As our case illustrates, it is crucially important for EM physicians to interpret non-contrast CT scans in a systematic and accurate manner. Clinical correlation is a distinct advantage that we, as emergency physicians, possess and it should be exploited to allow for timely and effective patient care.
Expert Commentary
Thanks to Drs. Jackson and Weygandt for this great primer to the emergent head CT. One of the obvious challenges of EM is the breadth of pathology we see, and so having a strategic approach like this one will reveal most of the emergent diagnoses we are looking for. I will never be a radiologist, but nothing is faster than looking at my own scan. A few thoughts: I start by scrolling a scan through quickly to identify obvious pathology (a bleed, midline shift, etc.) and then try to actively redirect my attention back to a systematic approach. It is easy to hone in on the obvious abnormality and miss smaller but crucial clues. Go through the same progression every time. Get comfortable with finding different windows for your imaging. If you only look in a brain window, you’ll miss critical diagnoses. Symmetry is your best friend - until it is not. We are remarkably good at picking out asymmetry when looking at imaging, which reveals many of the emergent diagnoses, but keep some of the symmetric processes in the back of your mind. Many of these can wait for a radiologist’s fine- tooth comb, but a few stand out. Get used to finding the basilar artery, particularly in your unconscious patient; an acute occlusion in this midline structure is potentially devastating but quick intervention is life-saving. Similarly, acute hydrocephalus merits immediate intervention that can lead to dramatic clinical improvement. Bilateral or midline subdural hemorrhage can also be easily missed; finding these requires a level of comfort with windowing the images and identifying abnormal CSF spaces.
Katie Colton, MD
Instructor, Feinberg School of Medicine
Department of Neuro Critical Care and Department of Emergency Medicine
Northwestern Memorial Hospital
How To Cite This Post:
[Peer-Reviewed, Web Publication] Philip, J. Weygandt, L. (2020, Feb 10). Non Contrast CT Head for the EM Physician. [NUEM Blog. Expert Commentary by Colton, K]. Retrieved from http://www.nuemblog.com/blog/non-contrast-ct-head-for-the-em-physician
Other Posts You May Enjoy
References
Adams, James, and Erik D. Barton. Emergency Medicine: Clinical Essentials. 2nd ed. N.p.: Elsevier Health Sciences, 2013;633-644.
Jamal K, Mandel L, Jamal L, Gilani S. 'Out of hours' adult CT head interpretation by senior emergency department staff following an intensive teaching session: a prospective blinded pilot study of 405 patients. Emergency medicine journal : EMJ. 2014;31(6):467-470.
Perron AD, Huff JS, Ullrich CG, Heafner MD, Kline JA. A multicenter study to improve emergency medicine residents' recognition of intracranial emergencies on computed tomography. Annals of emergency medicine. 1998;32(5):554-562.
Mayfield Brain & Spine. "Visual field test." Visual Field Test | Mayfield Brain & Spine. N.p., n.d. Web. 19 Dec. 2016.